Identification of a novel population of highly cytotoxic c-Met-expressing CD8+ T lymphocytes
- PMID: 28751311
- PMCID: PMC5579394
- DOI: 10.15252/embr.201744075
Identification of a novel population of highly cytotoxic c-Met-expressing CD8+ T lymphocytes
Abstract
CD8+ cytotoxic T lymphocytes (CTLs) are critical mediators of anti-tumor immunity, and controlling the mechanisms that govern CTL functions could be crucial for enhancing patient outcome. Previously, we reported that hepatocyte growth factor (HGF) limits effective murine CTL responses via antigen-presenting cells. Here, we show that a fraction of murine effector CTLs expresses the HGF receptor c-Met (c-Met+ CTLs). Phenotypic and functional analysis of c-Met+ CTLs reveals that they display enhanced cytolytic capacities compared to their c-Met- CTL counterparts. Furthermore, HGF directly restrains the cytolytic function of c-Met+ CTLs in cell-mediated cytotoxicity reactions in vitro and in vivo and abrogates T-cell responses against metastatic melanoma in vivo Finally, we establish in three murine tumor settings and in human melanoma tissues that c-Met+ CTLs are a naturally occurring CD8+ T-cell population. Together, our findings suggest that the HGF/c-Met pathway could be exploited to control CD8+ T-cell-mediated anti-tumor immunity.
Keywords: CTL; HGF; cancer; c‐Met; tumor immunity.
© 2017 The Authors.
Figures

Undifferentiated (day 0) and bulk day 5 differentiated live (7AAD−) Pmel‐1 TCR Tg CD3+ CD8+ T cells were analyzed, and mean percentage of c‐Met+ cells is shown. Frequency values are the result above the fluorescence values obtained by isotype control antibodies. Data are representative of three independent experiments (n = 3 mice/group).
Frequencies of c‐Met‐expressing Pmel‐1 CD3+ CD8+ T cells were analyzed by flow cytometry from three experiments of n = 3 mice/group over the course of in vitro CTL differentiation. The values represent frequencies of c‐Met+ cells in the total expressing Pmel‐1 CD3+ CD8+ T lymphocytes. Error bars show mean ± SEM, ***P < 0.001 (unpaired, 2‐tailed Student's t‐test).
Protein expression levels of c‐Met in undifferentiated (day 0), bulk day 5 differentiated, and FACS‐sorted day 5 c‐Met+ and c‐Met− Pmel‐1 CD3+ CD8+ T cells, as measured by Western blot. Data are representative of three independent experiments of n = 3 mice/group.
Relative expression of c‐Met mRNA in undifferentiated (day 0) and bulk day 5 differentiated Pmel‐1 CD8+ T cells, and FACS‐sorted day 5 c‐Met+ and c‐Met− Pmel‐1 CD3+ CD8+ T cells as measured by qRT–PCR. Data are representative of three independent experiments of n = 3 mice/group carried out in triplicate. Error bars show mean ± SEM, ***P < 0.001 (unpaired, 2‐tailed Student's t‐test).
Representative plots (histogram) and average geometric MFI (bar graph) of GrB, perforin, TNF, IFN‐γ, FasL, CD8, LFA‐1, CD107a, CCR4, and CXCR3 in day 5 differentiated c‐Met+ and c‐Met− Pmel‐1 CD3+ CD8+ T cells from three experiments of n = 3 mice/group. Error bars show mean ± SEM, *P < 0.05; **P < 0.01 (unpaired, 2‐tailed Student's t‐test).
IFN‐γ (left panel) and TNF (right panel) levels were quantified by ELISA from day 5 differentiated c‐Met+ and c‐Met− Pmel‐1 CD3+ CD8+ T cells stimulated by anti‐CD3/CD28 antibodies for 48 h. Data represent three experiments of n = 3 mice/group shown as mean ± SEM, **P < 0.01; ***P < 0.001 (unpaired, 2‐tailed Student's t‐test).
In vitro cytolytic activity of day 5 differentiated c‐Met+ and c‐Met− Pmel‐1 CD3+ CD8+ T cells. Various effector/target (E/T) ratios were tested for killing of syngeneic EL‐4 cells pulsed with hgp10025–33 peptide (squares) or control OVA257–264 peptide (circles). Values represent mean ± SEM of nine mice per group, pooled data from three independent experiments with three mice per group in each experiment, carried out in triplicate. *P < 0.05 (unpaired, 2‐tailed Student's t‐test).
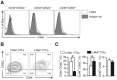
Representative flow cytometric histograms of c‐Met expression in splenic naïve (CD44loCD62Lhi), effector memory (CD44hiCD62Llo), and central memory (CD44hiCD62Lhi) Pmel‐1 CD3+ CD8+ T cells from healthy mice from one of three independent experiments (n = 3 mice/group).
Representative flow cytometric analysis of CD44 and CD62L expressions in day 5 differentiated c‐Met+ and c‐Met− Pmel‐1 CD3+ CD8+ T cells from one of three independent experiments of n = 3 mice/group. Percentages of cells in each quadrant are shown.
Tabulated results from (B) are presented as percentage of effector/memory in total CD3+ CD8+ T cells. Error bars show mean ± SEM, *P < 0.05 (unpaired, 2‐tailed Student's t‐test).

Representative plots (histogram) and average MFI (bar graph) of GrB, perforin, TNF, IFN‐γ, FasL, CD8, LFA‐1, CD107a, CCR4, and CXCR3 in day 5 differentiated c‐Met+ Pmel‐1 CD3+ CD8+ T cells treated with either vehicle (PBS) or HGF (30 ng/ml for 24 h) from three independent experiments of n = 3 mice/group. Error bars show mean ± SEM, *P < 0.05; **P < 0.01 (unpaired, 2‐tailed Student's t‐test).
Representative plots (histogram) and average geometric mean fluorescence intensity (MFI; bar graph) of GrB, perforin, TNF, IFN‐γ, FasL, CD8, LFA‐1, CD107a, CCR4, and CXCR3 in day 5 differentiated c‐Met− Pmel‐1 CD3+ CD8+ T cells treated with either vehicle or HGF (30 ng/ml for 24 h) from three independent experiments of n = 3 mice/group. Error bars show mean ± SEM (unpaired, 2‐tailed Student's t‐test).
IFN‐γ and TNF levels were quantified by ELISA from day 5 differentiated c‐Met+ and c‐Met− Pmel‐1 CD3+ CD8+ T cells treated with either vehicle or HGF (30 ng/ml for 24 h) and stimulated by anti‐CD3/CD28 antibodies for 48 h. Data represent three experiments of n = 3 mice/group shown as mean ± SEM, *P < 0.05; **P < 0.01; ***P < 0.001 (unpaired, 2‐tailed Student's t‐test).
In vitro cytolytic activity of day 5 differentiated c‐Met+ and c‐Met− Pmel‐1 CD3+ CD8+ T cells treated with either vehicle or HGF (30 ng/ml for 24 h). Various effector/target (E/T) ratios were tested for killing of syngeneic EL‐4 cells pulsed with hgp10025–33 peptide (squares) or control OVA257–264 peptide (circles). Values represent mean ± SEM of nine mice per group, pooled data from three independent experiments with three mice per group in each experiment, carried out in triplicate. *P < 0.05 (unpaired, 2‐tailed Student's t‐test).
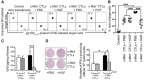
Representative dot plots of CFSE vs. BV profile of recovered CD45.1+ mouse splenocytes. Numbers indicate the percentage of cells of the high or null CFSE or BV phenotype.
Graphical representation (scatterplot) of analyzed data pooled from two experiments (n = 3 mice/group). Controls referred to as non‐transferred. Percentage of in vivo cytotoxic activity was calculated as described in Materials and Methods. Error bars show mean ± SEM, ***P < 0.001 (unpaired, 2‐tailed Student's t‐test).
Spleens were removed from each mouse at sacrifice, and content in GrB per gram of tissue was measured by ELISA. Error bars show mean ± SEM of 6 mice per group, ***P < 0.001 (unpaired, 2‐tailed Student's t‐test).
ELISpot assays were used to determine in individual mice the number of cells secreting IFN‐γ after ex vivo re‐stimulation of splenocytes with hgp10025–33 peptide for 24 h. Spots were counted with an ELISpot reader (Cellular Technology Ltd.) and expressed as spot‐forming cells per 1 × 106 splenocytes, after subtraction of control values (no peptide). Error bars show mean ± SEM, *P < 0.05 (unpaired, 2‐tailed Student's t‐test). A representative well image for each group is given on the left of the graph. Data are representative of two independent experiments (n = 3 mice/group).

- A
IFN‐γ and TNF levels from day 5 differentiated MACS‐separated bulk Pmel‐1 CD3+ CD8+ T cells treated with either vehicle or HGF (30 ng/ml for 24 h) and stimulated by anti‐CD3/CD28 antibodies for 48 h, quantified by ELISA. Data are shown as mean ± SEM of nine mice per group, pooled data from three individual experiments with three mice per group in each experiment, *P < 0.05 (unpaired, 2‐tailed Student's t‐test).
- B, C
Day 5 MACS‐separated bulk Pmel‐1 CTLs treated with either PBS or HGF (30 ng/ml for 24 h) were transferred i.v. into congenic CD45.2+ recipients. Equal parts of hgp10025–33‐pulsed brilliant violet (BV)‐labeled target CD45.1+ and unpulsed CFSE‐labeled CD45.1+ splenocytes were injected i.v. into recipient CD45.2+ mice 5 h after adoptive transfer of Pmel‐1 CTLs. Recipient CD45.2+ mice were sacrificed 4 h following transfer of target cells and control splenocytes, and the percentage of remaining target cells was quantified by FACS analysis. Representative (B) dot plots of BV vs. CFSE profile of recovered CD45.1+ mouse splenocytes. Numbers in histograms indicate the percentage of cells of the high or null BV or CFSE phenotype. (C) Graphical representation (scatterplot) of analyzed data from one of two independent experiments (n = 3 mice/group). Controls referred to as non‐transferred. Error bars show mean ± SEM, ***P < 0.001 (unpaired, 2‐tailed Student's t‐test).
- D
Representative plots (histogram) and average geometric MFI (bar graph) of c‐Met in day 5 differentiated c‐Met+ and c‐Met− Pmel‐1 CD3+ CD8+ T cells treated or not with either vehicle or HGF (30 ng/ml for 24 h). Composite data of three experiments with n = 3 mice/group is shown. Error bars show mean ± SEM.
- E
Relative expression of c‐Met mRNA in day 5 differentiated c‐Met+ and c‐Met− Pmel‐1 CD3+ CD8+ T cells treated or not with either vehicle or HGF (30 ng/ml for 24 h), as measured by qRT–PCR. Data are representative of three independent experiments (n = 3 mice/group) carried out in triplicate. Error bars show mean ± SEM.
- F
Frequencies of 7AAD+ c‐Met+ or c‐Met− CD8+ T cells were quantified by FACS on day 5 differentiated bulk Pmel‐1 CD3+ CD8+ T cells treated with either vehicle or HGF (30 ng/ml for 24 h). Data are representative of one of two independent experiments (n = 5 mice/group).
- G
In vitro cytolytic activity of day 5 differentiated OVA‐specific OT‐I CD3+ CD8+ T cells. Various effector/target (E/T) ratios were tested for killing of syngeneic EL‐4 cells pulsed with OVA257–264 peptide (squares) or control MOG44–54 peptide (circles). OVA‐specific OT‐I CD3+ CD8+ T cells were incubated a 1:5 ratio for 4 h prior to the assay with day 5 differentiated Pmel‐1 c‐Met+ CD3+ CD8+ T cells treated with either vehicle or HGF (30 ng/ml for 24 h). Values represent mean ± SEM of 9 mice per group, pooled data from three independent experiments with 3 mice per group in each experiment, carried out in triplicate. *P < 0.05 (unpaired, 2‐tailed Student's t‐test).
- H
GrB and IFN‐γ levels from supernatants recovered from the cytotoxic assays shown in (G) quantified by ELISA. Data are representative of three independent experiments (n = 3 mice/group) and are shown as mean ± SEM of triplicates, *P < 0.05 (unpaired, 2‐tailed Student's t‐test).
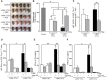
- A
Lung photograph from one of two independent experiments (n = 5 mice/group).
- B
Quantification of metastatic lung foci. Error bars show mean ± SEM of five mice per group, *P < 0.05 (unpaired, 2‐tailed Student's t‐test).
- C
Average MFI of KLRG1 levels by hgp10025–33 dextramer+ Thy1.1+ Pmel‐1 CD3+ CD8+ T cells found in lungs of mice were analyzed by flow cytometry from one of two experiments (n = 5 mice/group). Error bars show mean ± SEM, **P < 0.01 (unpaired, 2‐tailed Student's t‐test).
- D
Average MFI of IFN‐γ levels expressed by tumor‐infiltrating hgp10025–33 dextramer+ Thy1.1+ Pmel‐1 CD3+ CD8+ T cells after 6 h of stimulation with 1 μM hgp10025–33 peptide. Data represent one of two experiments (n = 5 mice/group). Error bars show mean ± SEM, *P < 0.05 (unpaired, 2‐tailed Student's t‐test).
- E, F
IFN‐γ (E) and GrB (F) levels were quantified by ELISA in the cell supernatant of tumor‐infiltrating T cells (TILs) after 6 h of stimulation with 1 μM hgp10025–33 peptide. Data represent one of two experiments (n = 5 mice/group) shown as mean ± SEM, *P < 0.05; **P < 0.01 (unpaired, 2‐tailed Student's t‐test).

- A–C
Representative plots (contour plots) and mean frequencies (bar graphs) of hgp10025–33 dextramer+ Thy1.1+ Pmel‐1 CD3+ CD8+ T cells found in (A) lungs, (B) DLNs, and (C) spleens of individual mice adoptively transferred with Thy1.1+ Pmel‐1 CTLs. Frequency values are the result above the fluorescence values obtained by isotype control antibodies as measured by flow cytometry. The values represent frequencies of hgp10025–33 dextramer+ cells in the total Thy1.1+ Pmel‐1 CD3+ CD8+ T lymphocytes. Controls are referred to as non‐transferred. Data are representative of one of two independent experiments (n = 5 mice/group). Error bars show mean ± SEM.
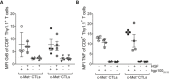
- A, B
Average MFI of (A) Grb and (B) TNF levels expressed by hgp10025–33 dextramer+ Thy1.1+ Pmel‐1 CD3+ CD8+ T cells found in spleens of mice after 6 h of stimulation with 1 μM hgp10025–33 peptide or an irrelevant control Ag (OVA257–264). Data represent one of two experiments (n = 4 mice/group). Error bars show mean ± SEM.
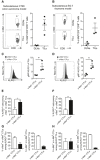
- A, B
Representative contour plots and mean frequencies (scatterplots) of c‐Met+ tumor‐DLN‐resident CD3+ CD8+ T lymphocytes and tumor‐infiltrating CD3+ CD8+ T lymphocytes (TILs) of mice bearing subcutaneous CT26 carcinomas or EG‐7 thymomas. Frequency values are the result above the fluorescence values obtained by isotype control antibodies as measured by flow cytometry. The values represent frequencies of c‐Met+ cells in the total CD3+ CD8+ T lymphocytes. Data are representative of one of two independent experiments (n = 3 mice/group). Errors bars show mean ± SEM.
- C, D
Representative plots (histograms) and average MFI (scatterplots) of KLRG1 expression on tumor‐infiltrating c‐Met+ or c‐Met− CD3+ CD8+ T lymphocytes of mice bearing subcutaneous CT26 carcinomas or EG‐7 thymomas. Results are presented as MFI after normalizing the values with the isotype control. Error bars show mean ± SEM, *P < 0.05 (unpaired, 2‐tailed Student's t‐test); data are representative of one of two independent experiments (n = 3 mice/group).
- E, F
Mean frequencies (bar graphs) of KLRG1+ cells among c‐Met+ or c‐Met− TILs of mice bearing subcutaneous CT26 carcinomas or EG‐7 thymomas. Data represent one of two experiments (n = 3 mice/group) shown as mean ± SEM, **P < 0.01 (unpaired, 2‐tailed Student's t‐test).
- G, H
Mean frequencies (bar graphs) of c‐Met+ or c‐Met− cells among KLRG1+ (left panel) or KLRG1− (right panel) TILs of mice bearing subcutaneous CT26 carcinomas or EG‐7 thymomas. Data represent one of two experiments (n = 3 mice/group) shown as mean ± SEM (unpaired, 2‐tailed Student's t‐test).
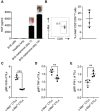
- A
HGF protein expression in tumors produced by B16 melanoma cells was determined by ELISA analysis after 7 or 14 days of transplantation of these cancer cells. HGF protein expression in B16 melanoma cells was also determined (n = 3 lungs/group). HGF was expressed in B16 tumors, but not detectable in B16 cells or healthy lung. Data shown are means ± SEM from one of two independent experiments. Representative photographs of tumor‐bearing lungs are shown.
- B
Representative density plot and mean frequency (scatterplot) of c‐Met+ tumor‐infiltrating CD3+ CD8+ T lymphocytes (TILs) of mice bearing B16 lung melanoma tumors (day 14). Frequency values are the result above the fluorescence values obtained by isotype control antibodies as measured by flow cytometry. The values represent frequencies of c‐Met+ cells in the total CD3+ CD8+ T lymphocytes. Data are representative of one of two independent experiments (n = 4 mice/group) and are shown as means ± SEM.
- C–E
Average geometric mean fluorescence intensity (MFI) (scatterplot) of (C) GrB, (D) TNF, and (E) IFN‐γ in CD3+ CD8+ T cells found in lungs of mice. Data shown are means ± SEM from one of two independent experiments (n = 4 mice/group), **P < 0.01; ***P < 0.001 (unpaired, 2‐tailed Student's t‐test).
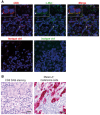
Representative double immunofluorescent staining images for CD8 (red) and c‐Met (green) of sections of human malignant melanoma tissues at ×40 magnification. Cell nuclei were counterstained with DAPI (blue). Scale bar, 20 μm. Insets are magnified 4.5× from the original images. The asterisk indicates the area used to acquire the images shown in inset. Comparable results were obtained with three other patients.
Representative DAB staining of tumor samples showing melanoma cells surrounded by CD8+ T lymphocytes (left). Brown staining from the DAB indicates CD8+ T cells. Representative Melan‐A immunohistochemistry of melanoma cells stained in red (right). Scale bar, 20 μm. Comparable results were obtained with three other patients.
Similar articles
-
c-Met+ Cytotoxic T Lymphocytes Exhibit Enhanced Cytotoxicity in Mice and Humans In Vitro Tumor Models.Biomedicines. 2023 Nov 23;11(12):3123. doi: 10.3390/biomedicines11123123. Biomedicines. 2023. PMID: 38137344 Free PMC article.
-
c-Met is expressed by highly autoreactive encephalitogenic CD8+ cells.J Neuroinflammation. 2020 Feb 19;17(1):68. doi: 10.1186/s12974-019-1676-0. J Neuroinflammation. 2020. PMID: 32075650 Free PMC article.
-
The neurotrophic hepatocyte growth factor attenuates CD8+ cytotoxic T-lymphocyte activity.J Neuroinflammation. 2013 Dec 17;10:154. doi: 10.1186/1742-2094-10-154. J Neuroinflammation. 2013. PMID: 24344806 Free PMC article.
-
Molecular mechanisms and biological significance of CTL avidity.Curr HIV Res. 2003 Jul;1(3):287-94. doi: 10.2174/1570162033485230. Curr HIV Res. 2003. PMID: 15046253 Review.
-
CD8+ cytotoxic T lymphocytes in cancer immunotherapy: A review.J Cell Physiol. 2019 Jun;234(6):8509-8521. doi: 10.1002/jcp.27782. Epub 2018 Nov 22. J Cell Physiol. 2019. PMID: 30520029 Review.
Cited by
-
T-Cell Subpopulations Exhibit Distinct Recruitment Potential, Immunoregulatory Profile and Functional Characteristics in Chagas versus Idiopathic Dilated Cardiomyopathies.Front Cardiovasc Med. 2022 Feb 2;9:787423. doi: 10.3389/fcvm.2022.787423. eCollection 2022. Front Cardiovasc Med. 2022. PMID: 35187122 Free PMC article.
-
Circulating c-Met-Expressing Memory T Cells Define Cardiac Autoimmunity.Circulation. 2022 Dec 20;146(25):1930-1945. doi: 10.1161/CIRCULATIONAHA.121.055610. Epub 2022 Nov 23. Circulation. 2022. PMID: 36417924 Free PMC article.
-
Phase I Study of Ficlatuzumab and Cetuximab in Cetuximab-Resistant, Recurrent/Metastatic Head and Neck Cancer.Cancers (Basel). 2020 Jun 11;12(6):1537. doi: 10.3390/cancers12061537. Cancers (Basel). 2020. PMID: 32545260 Free PMC article.
-
c-Met+ Cytotoxic T Lymphocytes Exhibit Enhanced Cytotoxicity in Mice and Humans In Vitro Tumor Models.Biomedicines. 2023 Nov 23;11(12):3123. doi: 10.3390/biomedicines11123123. Biomedicines. 2023. PMID: 38137344 Free PMC article.
-
GD2-directed CAR-T cells in combination with HGF-targeted neutralizing antibody (AMG102) prevent primary tumor growth and metastasis in Ewing sarcoma.Int J Cancer. 2020 Jun 1;146(11):3184-3195. doi: 10.1002/ijc.32743. Epub 2019 Nov 6. Int J Cancer. 2020. PMID: 31621900 Free PMC article.
References
-
- Zhang YW, Vande Woude GF (2003) HGF/SF‐met signaling in the control of branching morphogenesis and invasion. J Cell Biochem 88: 408–417 - PubMed
-
- Birchmeier C, Gherardi E (1998) Developmental roles of HGF/SF and its receptor, the c‐Met tyrosine kinase. Trends Cell Biol 8: 404–410 - PubMed
-
- Trusolino L, Bertotti A, Comoglio PM (2001) A signaling adapter function for alpha6beta4 integrin in the control of HGF‐dependent invasive growth. Cell 107: 643–654 - PubMed
-
- Hellstrom KE, Hellstrom I (1969) Cellular immunity against tumor antigens. Adv Cancer Res 12: 167–223 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

