The vascular basement membrane in the healthy and pathological brain
- PMID: 28753105
- PMCID: PMC5624399
- DOI: 10.1177/0271678X17722436
The vascular basement membrane in the healthy and pathological brain
Abstract
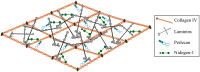
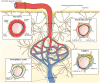
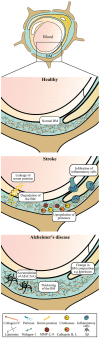
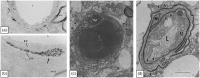
The vascular basement membrane contributes to the integrity of the blood-brain barrier (BBB), which is formed by brain capillary endothelial cells (BCECs). The BCECs receive support from pericytes embedded in the vascular basement membrane and from astrocyte endfeet. The vascular basement membrane forms a three-dimensional protein network predominantly composed of laminin, collagen IV, nidogen, and heparan sulfate proteoglycans that mutually support interactions between BCECs, pericytes, and astrocytes. Major changes in the molecular composition of the vascular basement membrane are observed in acute and chronic neuropathological settings. In the present review, we cover the significance of the vascular basement membrane in the healthy and pathological brain. In stroke, loss of BBB integrity is accompanied by upregulation of proteolytic enzymes and degradation of vascular basement membrane proteins. There is yet no causal relationship between expression or activity of matrix proteases and the degradation of vascular matrix proteins in vivo. In Alzheimer's disease, changes in the vascular basement membrane include accumulation of Aβ, composite changes, and thickening. The physical properties of the vascular basement membrane carry the potential of obstructing drug delivery to the brain, e.g. thickening of the basement membrane can affect drug delivery to the brain, especially the delivery of nanoparticles.
Keywords: Alzheimer’s disease; blood-brain barrier; drug delivery; stroke; the brain’s vascular basement membrane.
Figures
References
-
- Paulsson M. Basement membrane proteins: structure, assembly, and cellular interactions. Crit Rev Biochem Mol Biol 1992; 27: 93–127. - PubMed
-
- Abbott NJ, Ronnback L, Hansson E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat Rev Neurosci 2006; 7: 41–53. - PubMed
-
- Lichota J, Skjorringe T, Thomsen LB, et al. Macromolecular drug transport into the brain using targeted therapy. J Neurochem 2010; 113: 1–13. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

