In Vivo Roles of Fatty Acid Biosynthesis Enzymes in Biosynthesis of Biotin and α-Lipoic Acid in Corynebacterium glutamicum
- PMID: 28754705
- PMCID: PMC5601351
- DOI: 10.1128/AEM.01322-17
In Vivo Roles of Fatty Acid Biosynthesis Enzymes in Biosynthesis of Biotin and α-Lipoic Acid in Corynebacterium glutamicum
Abstract
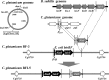
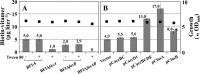
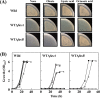
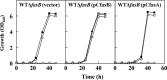
For fatty acid biosynthesis, Corynebacterium glutamicum uses two type I fatty acid synthases (FAS-I), FasA and FasB, in addition to acetyl-coenzyme A (CoA) carboxylase (ACC) consisting of AccBC, AccD1, and AccE. The in vivo roles of the enzymes in supplying precursors for biotin and α-lipoic acid remain unclear. Here, we report genetic evidence demonstrating that the biosynthesis of these cofactors is linked to fatty acid biosynthesis through the FAS-I pathway. For this study, we used wild-type C. glutamicum and its derived biotin vitamer producer BFI-5, which was engineered to express Escherichia coli bioBF and Bacillus subtilis bioI Disruption of either fasA or fasB in strain BFI-5 led to decreased production of biotin vitamers, whereas its amplification contributed to increased production, with a larger impact of fasA in both cases. Double disruptions of fasA and fasB resulted in no biotin vitamer production. The acc genes showed a positive effect on production when amplified simultaneously. Augmented fatty acid biosynthesis was also reflected in pimelic acid production when carbon flow was blocked at the BioF reaction. These results indicate that carbon flow down the FAS-I pathway is destined for channeling into the biotin biosynthesis pathway, and that FasA in particular has a significant impact on precursor supply. In contrast, fasB disruption resulted in auxotrophy for lipoic acid or its precursor octanoic acid in both wild-type and BFI-5 strains. The phenotypes were fully complemented by plasmid-mediated expression of fasB but not fasA These results reveal that FasB plays a specific physiological role in lipoic acid biosynthesis in C. glutamicumIMPORTANCE For the de novo biosynthesis of fatty acids, C. glutamicum exceptionally uses a eukaryotic multifunctional type I fatty acid synthase (FAS-I) system comprising FasA and FasB, in contrast to most bacteria, such as E. coli and B. subtilis, which use an individual nonaggregating type II fatty acid synthase (FAS-II) system. In this study, we reported genetic evidence demonstrating that the FAS-I system is the source of the biotin precursor in vivo in the engineered biotin-prototrophic C. glutamicum strain. This study also uncovered the important physiological role of FasB in lipoic acid biosynthesis. Here, we present an FAS-I enzyme that functions in supplying the lipoic acid precursor, although its biosynthesis has been believed to exclusively depend on FAS-II in organisms. The findings obtained here provide new insights into the metabolic engineering of this industrially important microorganism to produce these compounds effectively.
Keywords: Corynebacterium glutamicum; biotin; fatty acid biosynthesis; lipoic acid; metabolic engineering.
Copyright © 2017 American Society for Microbiology.
Figures

Similar articles
-
Two functional FAS-I type fatty acid synthases in Corynebacterium glutamicum.Microbiology (Reading). 2005 Jul;151(Pt 7):2421-2427. doi: 10.1099/mic.0.28012-0. Microbiology (Reading). 2005. PMID: 16000732
-
The TetR-type transcriptional regulator FasR of Corynebacterium glutamicum controls genes of lipid synthesis during growth on acetate.Mol Microbiol. 2010 Oct;78(1):253-65. doi: 10.1111/j.1365-2958.2010.07337.x. Mol Microbiol. 2010. PMID: 20923423
-
Development of biotin-prototrophic and -hyperauxotrophic Corynebacterium glutamicum strains.Appl Environ Microbiol. 2013 Aug;79(15):4586-94. doi: 10.1128/AEM.00828-13. Epub 2013 May 24. Appl Environ Microbiol. 2013. PMID: 23709504 Free PMC article.
-
Biosynthesis of biotin and lipoic acid.Vitam Horm. 2001;61:51-101. doi: 10.1016/s0083-6729(01)61002-1. Vitam Horm. 2001. PMID: 11153271 Review.
-
Metabolic engineering of Corynebacterium glutamicum for producing branched chain amino acids.Microb Cell Fact. 2021 Dec 24;20(1):230. doi: 10.1186/s12934-021-01721-0. Microb Cell Fact. 2021. PMID: 34952576 Free PMC article. Review.
Cited by
-
Nutrient Effects on Motor Neurons and the Risk of Amyotrophic Lateral Sclerosis.Nutrients. 2021 Oct 26;13(11):3804. doi: 10.3390/nu13113804. Nutrients. 2021. PMID: 34836059 Free PMC article. Review.
-
Metabolism of Dietary and Microbial Vitamin B Family in the Regulation of Host Immunity.Front Nutr. 2019 Apr 17;6:48. doi: 10.3389/fnut.2019.00048. eCollection 2019. Front Nutr. 2019. PMID: 31058161 Free PMC article. Review.
-
Beyond rational-biosensor-guided isolation of 100 independently evolved bacterial strain variants and comparative analysis of their genomes.BMC Biol. 2023 Sep 4;21(1):183. doi: 10.1186/s12915-023-01688-x. BMC Biol. 2023. PMID: 37667306 Free PMC article.
-
A Futile Metabolic Cycle of Fatty Acyl-CoA Hydrolysis and Resynthesis in Corynebacterium glutamicum and Its Disruption Leading to Fatty Acid Production.Appl Environ Microbiol. 2021 Mar 1;87(5):e02469-20. doi: 10.1128/AEM.02469-20. Epub 2020 Dec 11. Appl Environ Microbiol. 2021. PMID: 33310719 Free PMC article.
-
A Canonical Biotin Synthesis Enzyme, 8-Amino-7-Oxononanoate Synthase (BioF), Utilizes Different Acyl Chain Donors in Bacillus subtilis and Escherichia coli.Appl Environ Microbiol. 2017 Dec 15;84(1):e02084-17. doi: 10.1128/AEM.02084-17. Print 2018 Jan 1. Appl Environ Microbiol. 2017. PMID: 29054876 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

