Genetic and neuronal mechanisms governing the sex-specific interaction between sleep and sexual behaviors in Drosophila
- PMID: 28754889
- PMCID: PMC5533705
- DOI: 10.1038/s41467-017-00087-5
Genetic and neuronal mechanisms governing the sex-specific interaction between sleep and sexual behaviors in Drosophila
Abstract
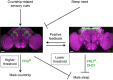
Animals execute one particular behavior among many others in a context-dependent manner, yet the mechanisms underlying such behavioral choice remain poorly understood. Here we studied how two fundamental behaviors, sex and sleep, interact at genetic and neuronal levels in Drosophila. We show that an increased need for sleep inhibits male sexual behavior by decreasing the activity of the male-specific P1 neurons that coexpress the sex determination genes fru M and dsx, but does not affect female sexual behavior. Further, we delineate a sex-specific neuronal circuit wherein the P1 neurons encoding increased courtship drive suppressed male sleep by forming mutually excitatory connections with the fru M -positive sleep-controlling DN1 neurons. In addition, we find that FRUM regulates male courtship and sleep through distinct neural substrates. These studies reveal the genetic and neuronal basis underlying the sex-specific interaction between sleep and sexual behaviors in Drosophila, and provide insights into how competing behaviors are co-regulated.Genes and circuits involved in sleep and sexual arousal have been extensively studied in Drosophila. Here the authors identify the sex determination genes fruitless and doublesex, and a sex-specific P1-DN1 neuronal feedback that governs the interaction between these competing behaviors.
Conflict of interest statement
The authors declare no competing financial interests.
Figures
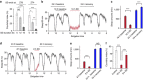


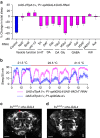
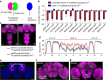
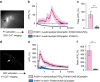
References
-
- McFarland DJ. Decision making in animals. Nature. 1977;269:15–21. doi: 10.1038/269015a0. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

