New prodrugs of two pyrimidine acyclic nucleoside phosphonates: Synthesis and antiviral activity
- PMID: 28757102
- PMCID: PMC7126465
- DOI: 10.1016/j.bmc.2017.06.046
New prodrugs of two pyrimidine acyclic nucleoside phosphonates: Synthesis and antiviral activity
Abstract
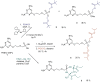
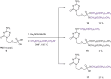
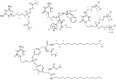
New 2,4-diamino-6-[2-(phosphonomethoxy)ethoxy]pyrimidine (PMEO-DAPy) and 1-[2-(phosphonomethoxy)ethyl]-5-azacytosine (PME-5-azaC) prodrugs were prepared with a pro-moiety consisting of carbonyloxymethyl esters (POM, POC), alkoxyalkyl esters, amino acid phosphoramidates and/or tyrosine. The activity of the prodrugs was evaluated in vitro against different virus families. None of the synthesized prodrugs demonstrated activity against RNA viruses but some of them proved active against herpesviruses [including herpes simplex virus (HSV), varicella-zoster virus (VZV), and human cytomegalovirus (HCMV)]. The bis(POC) and the bis(amino acid) phosphoramidate prodrugs of PMEO-DAPy inhibited herpesvirus replication at lower doses than the parent compound although the selectivity against HSV and VZV was only slightly improved compared to PMEO-DAPy. The mono-octadecyl ester of PME-5-azaC emerged as the most potent and selective PME-5-azaC prodrug against HSV, VZV and HCMV with EC50's of 0.15-1.12µM while PME-5-azaC only had marginal anti-herpesvirus activity. Although the bis(hexadecylamido-l-tyrosyl) and the bis(POM) esters of PME-5-azaC were also very potent anti-herpesvirus drugs, these were less selective than the mono-octadecyl ester prodrug.
Keywords: 5-Azacytosine; Acyclic nucleoside phosphonates; Antivirals; HPMP-5-azaC; Open-ring; PME-azaC; PMEO-DAPy; Phosphonate; Prodrug.
Copyright © 2017 Elsevier Ltd. All rights reserved.
Figures
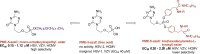





Similar articles
-
Phosphonates and Phosphonate Prodrugs in Medicinal Chemistry: Past Successes and Future Prospects.Front Chem. 2022 May 20;10:889737. doi: 10.3389/fchem.2022.889737. eCollection 2022. Front Chem. 2022. PMID: 35668826 Free PMC article. Review.
-
N(4)-Acyl derivatives as lipophilic prodrugs of cidofovir and its 5-azacytosine analogue, (S)-HPMP-5-azaC: chemistry and antiviral activity.Bioorg Med Chem. 2014 May 15;22(10):2896-906. doi: 10.1016/j.bmc.2014.03.031. Epub 2014 Mar 26. Bioorg Med Chem. 2014. PMID: 24731540
-
Synthesis of LAVR-289, a new [(Z)-3-(acetoxymethyl)-4-(2,4-diaminopyrimidin-6-yl)oxy-but-2-enyl]phosphonic acid prodrug with pronounced antiviral activity against DNA viruses.Eur J Med Chem. 2024 May 5;271:116412. doi: 10.1016/j.ejmech.2024.116412. Epub 2024 Apr 15. Eur J Med Chem. 2024. PMID: 38643669
-
Expanding the Antiviral Spectrum of 3-Fluoro-2-(phosphonomethoxy)propyl Acyclic Nucleoside Phosphonates: Diamyl Aspartate Amidate Prodrugs.J Med Chem. 2017 Jul 27;60(14):6220-6238. doi: 10.1021/acs.jmedchem.7b00416. Epub 2017 Jul 6. J Med Chem. 2017. PMID: 28682067
-
Amino Acid Ester Prodrugs of Nucleoside and Nucleotide Antivirals.Mini Rev Med Chem. 2017;17(10):818-833. doi: 10.2174/1389557517666170216151601. Mini Rev Med Chem. 2017. PMID: 28215138 Review.
Cited by
-
Novel camphor-based pyrimidine derivatives induced cancer cell death through a ROS-mediated mitochondrial apoptosis pathway.RSC Adv. 2019 Sep 19;9(51):29711-29720. doi: 10.1039/c9ra05900h. eCollection 2019 Sep 18. RSC Adv. 2019. PMID: 35531556 Free PMC article.
-
COVID-19 therapy: What weapons do we bring into battle?Bioorg Med Chem. 2020 Dec 1;28(23):115757. doi: 10.1016/j.bmc.2020.115757. Epub 2020 Sep 10. Bioorg Med Chem. 2020. PMID: 32992245 Free PMC article.
-
Advances and Perspectives in the Management of Varicella-Zoster Virus Infections.Molecules. 2021 Feb 20;26(4):1132. doi: 10.3390/molecules26041132. Molecules. 2021. PMID: 33672709 Free PMC article. Review.
-
Synthesis of fluorinated acyclic nucleoside phosphonates with 5-azacytosine base moiety.Tetrahedron. 2019 Sep 27;75(39):130529. doi: 10.1016/j.tet.2019.130529. Epub 2019 Aug 16. Tetrahedron. 2019. PMID: 32287433 Free PMC article.
-
Phosphonates and Phosphonate Prodrugs in Medicinal Chemistry: Past Successes and Future Prospects.Front Chem. 2022 May 20;10:889737. doi: 10.3389/fchem.2022.889737. eCollection 2022. Front Chem. 2022. PMID: 35668826 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

