Light driven mesoscale assembly of a coordination polymeric gelator into flowers and stars with distinct properties
- PMID: 28757961
- PMCID: PMC5506632
- DOI: 10.1039/c5sc02233a
Light driven mesoscale assembly of a coordination polymeric gelator into flowers and stars with distinct properties
Abstract
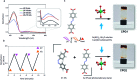
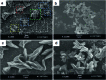
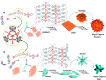
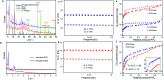
Control over the self-assembly process of porous organic-inorganic hybrids often leads to unprecedented polymorphism and properties. Herein we demonstrate how light can be a powerful tool to intervene in the kinetically controlled mesoscale self-assembly of a coordination polymeric gelator. Ultraviolet light induced coordination modulation via photoisomerisation of an azobenzene based dicarboxylate linker followed by aggregation mediated crystal growth resulted in two distinct morphological forms (flowers and stars), which show subtle differences in their physical properties.
Figures


Similar articles
-
Kinetic Control of Interpenetration in Fe-Biphenyl-4,4'-dicarboxylate Metal-Organic Frameworks by Coordination and Oxidation Modulation.J Am Chem Soc. 2019 May 22;141(20):8346-8357. doi: 10.1021/jacs.9b03269. Epub 2019 May 8. J Am Chem Soc. 2019. PMID: 31017428
-
Modular design of domain assembly in porous coordination polymer crystals via reactivity-directed crystallization process.J Am Chem Soc. 2012 Aug 15;134(32):13341-7. doi: 10.1021/ja303588m. Epub 2012 Aug 6. J Am Chem Soc. 2012. PMID: 22816727
-
Photoisomerisation and light-induced morphological switching of a polyoxometalate-azobenzene hybrid.Chem Commun (Camb). 2017 Jun 29;53(53):7278-7281. doi: 10.1039/c7cc01805c. Chem Commun (Camb). 2017. PMID: 28425541
-
Fluorescent Supramolecular Polymeric Materials.Adv Mater. 2017 Apr;29(14). doi: 10.1002/adma.201606117. Epub 2017 Feb 15. Adv Mater. 2017. PMID: 28198107 Review.
-
Reversible Phase Transition of Porous Coordination Polymers.Chemistry. 2020 Mar 2;26(13):2766-2779. doi: 10.1002/chem.201903985. Epub 2020 Jan 16. Chemistry. 2020. PMID: 31697441 Review.
Cited by
-
Innovative Microstructural Transformation upon CO2 Supercritical Conditions on Metal-Nucleobase Aerogel and Its Use as Effective Filler for HPLC Biomolecules Separation.Nanomaterials (Basel). 2022 Feb 17;12(4):675. doi: 10.3390/nano12040675. Nanomaterials (Basel). 2022. PMID: 35215003 Free PMC article.
-
A π-gel scaffold for assembling fullerene to photoconducting supramolecular rods.Sci Adv. 2016 Sep 23;2(9):e1600142. doi: 10.1126/sciadv.1600142. eCollection 2016 Sep. Sci Adv. 2016. PMID: 27679815 Free PMC article.
-
Supramolecular organogels fabricated with dicarboxylic acids and primary alkyl amines: controllable self-assembled structures.RSC Adv. 2020 Aug 6;10(49):29129-29138. doi: 10.1039/d0ra05072e. eCollection 2020 Aug 5. RSC Adv. 2020. PMID: 35521101 Free PMC article.
-
Probing E/Z Isomerism Using Pillar[4]pyridinium/Gold Nanoparticle Ensembles and Their Photoresponsive Behavior.Langmuir. 2022 Apr 26;38(16):4942-4947. doi: 10.1021/acs.langmuir.2c00342. Epub 2022 Apr 15. Langmuir. 2022. PMID: 35426683 Free PMC article.
-
Stepwise control of host-guest interaction using a coordination polymer gel.Nat Commun. 2018 May 18;9(1):1987. doi: 10.1038/s41467-018-04303-8. Nat Commun. 2018. PMID: 29777098 Free PMC article.
References
-
- Aono M. Sci. Technol. Adv. Mater. 2011;12:040301. - PMC - PubMed
- Ariga K., Ji Q., Hill J. P., Bando Y., Aono M. NPG Asia Mater. 2012;4:e17.
- Ariga K., Ji Q., Nakanishi W., Hill J. P., Aono M. Mater. Horiz. 2015;2:406.
- Ariga K., Yamauchi Y., Rydzek G., Ji Q., Yonamine Y., Wu K. C.-W., Hill J. P. Chem. Lett. 2014;43:36.
-
- Li H., Eddaoudi M., O'Keeffe M., Yaghi O. M. Nature. 1999;402:276.
- Furukawa H., Cordova K. E., O'Keeffe M., Yaghi O. M. Science. 2013;341:1230444. - PubMed
-
- Kondo M., Yoshitomi T., Seki K., Matsuzaka H., Kitagawa S. Angew. Chem., Int. Ed. Engl. 1997;36:1725.
- Kitagawa S., Kitaura R., Noro S. Angew. Chem., Int. Ed. 2004;43:2334. - PubMed
-
- Kishi N., Akita M., Kamiya M., Hayashi S., Hsu H.-F., Yoshizawa M. J. Am. Chem. Soc. 2013;135:12976. - PubMed
- Mukhopadhyay R. D., Praveen V. K., Ajayaghosh A. Mater. Horiz. 2014;1:572.
-
- Gopal A., Hifsudheen M., Furumi S., Takeuchi M., Ajayaghosh A. Angew. Chem., Int. Ed. 2012;51:10505. - PubMed
- Mahesh S., Gopal A., Thirumalai R., Ajayaghosh A. J. Am. Chem. Soc. 2012;134:7227. - PubMed
- Rajaganesh R., Gopal A., Das T. M., Ajayaghosh A. Org. Lett. 2012;14:748. - PubMed
- Abellán G., Coronado E., Martí-Gastaldo C., Ribera A., Jordá J. L., García H. Adv. Mater. 2014;26:4156. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

