Naphthalenebisimides as photofunctional surfactants for SWCNTs - towards water-soluble electron donor-acceptor hybrids
- PMID: 28757977
- PMCID: PMC5510015
- DOI: 10.1039/c5sc02944a
Naphthalenebisimides as photofunctional surfactants for SWCNTs - towards water-soluble electron donor-acceptor hybrids
Abstract
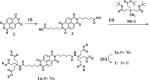
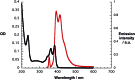
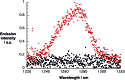
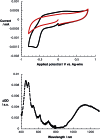
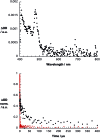
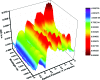
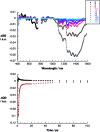
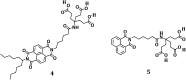
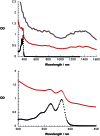
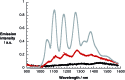
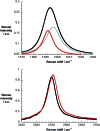
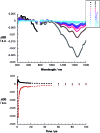
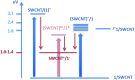
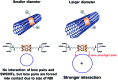
A water soluble naphthalenebisimide derivative (NBI) was synthesized and probed to individualize, suspend, and stabilize single wall carbon nanotubes (SWCNTs). Besides a comprehensive photophysical and electrochemical characterization of NBI, stable suspensions of SWCNTs were realized in buffered D2O. Overall, the dispersion efficiency of the NBI surfactant was determined by comparison with naphthalene based references. Successful individualization of SWCNTs was corroborated in several microscopic assays. In addition, emission spectroscopy points to the strong quenching of SWCNT centered band gap emission, when NBIs are immobilized onto SWCNTs. The origin of the quenching was found to be strong electronic communication, which leads to charge separation between NBIs and photoexcited SWCNTs, and, which yields reduced NBIs as well oxidized SWCNTs. Notably, electrochemical considerations revealed that the energy content of these charge separated states is one of the highest reported for SWCNT based electron donor-acceptor hybrids so far.
Figures

References
-
- Iijima S. Nature. 1991;354:56.
-
- Wang Z., Mohammadzadeh S., Schmaltz T., Kirschner J., Khassanov A., Eigler S., Mundloch U., Backes C., Steinrück H.-G., Magerl A., Hauke F., Hirsch A., Halik M. ACS Nano. 2013;7:11427. - PubMed
- Avouris P., Chen Z., Perebeinos V. Nat. Nanotechnol. 2007;2:605. - PubMed
- Park H., Afzali A., Han S.-J., Tulevski G. S., Franklin A. D., Tersoff J., Hannon J. B., Haensch W. Nat. Nanotechnol. 2012;7:787. - PubMed
-
- Toma F. M., Sartorel A., Iurlo M., Carraro M., Parisse P., Maccato C., Rapino S., Rodriguez Gonzalez B., Amenitsch H., Da Ros T., Casalis L., Goldoni A., Marcaccio M., Scorrano G., Scoles G., Paolucci F., Prato M., Bonchio M. Nat. Chem. 2010;2:826. - PubMed
-
- Bartelmess J., Ballesteros B., de la Torre G., Kiessling D., Campidelli S., Prato M., Torres T., Guldi D. M. J. Am. Chem. Soc. 2010;132:16202. - PubMed
- Campidelli S., Ballesteros B., Filoramo A., Diaz D. D., de la Torre G., Torres T., Rahman G. M. A., Ehli C., Kiessling D., Werner F., Sgobba V., Guldi D. M., Cioffi C., Prato M., Bourgoin J.-P. J. Am. Chem. Soc. 2008;130:11503. - PubMed
- Ince M., Bartelmess J., Kiessling D., Dirian K., Martınez-Diaz M. V., Torres T., Guldi D. M. Chem. Sci. 2012;3:1472.
- Guldi D. M., Sgobba V. Chem. Commun. 2011;47:606. - PubMed
- Sandanayaka A. S. D., Maligaspe E., Hasobe T., Ito O., D'Souza F. Chem. Commun. 2010;46:8749. - PubMed
- Das S. K., Subbaiyan N. K., D'Souza F., Sandanayaka A. S. D., Hasobe T., Ito O. Energy Environ. Sci. 2011;4:707.
- Dirian K., Herranz A. M., Katsukis G., Malig J., Rodriguez-Perez L., Romero-Nieto C., Strauss V., Martin N., Guldi D. M. Chem. Sci. 2013;4:4335.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

