Hyperpolarized 13C MR metabolic imaging can detect neuroinflammation in vivo in a multiple sclerosis murine model
- PMID: 28760957
- PMCID: PMC5565402
- DOI: 10.1073/pnas.1613345114
Hyperpolarized 13C MR metabolic imaging can detect neuroinflammation in vivo in a multiple sclerosis murine model
Abstract
Proinflammatory mononuclear phagocytes (MPs) play a crucial role in the progression of multiple sclerosis (MS) and other neurodegenerative diseases. Despite advances in neuroimaging, there are currently limited available methods enabling noninvasive detection of MPs in vivo. Interestingly, upon activation and subsequent differentiation toward a proinflammatory phenotype MPs undergo metabolic reprogramming that results in increased glycolysis and production of lactate. Hyperpolarized (HP) 13C magnetic resonance spectroscopic imaging (MRSI) is a clinically translatable imaging method that allows noninvasive monitoring of metabolic pathways in real time. This method has proven highly useful to monitor the Warburg effect in cancer, through MR detection of increased HP [1-13C]pyruvate-to-lactate conversion. However, to date, this method has never been applied to the study of neuroinflammation. Here, we questioned the potential of 13C MRSI of HP [1-13C]pyruvate to monitor the presence of neuroinflammatory lesions in vivo in the cuprizone mouse model of MS. First, we demonstrated that 13C MRSI could detect a significant increase in HP [1-13C]pyruvate-to-lactate conversion, which was associated with a high density of proinflammatory MPs. We further demonstrated that the increase in HP [1-13C]lactate was likely mediated by pyruvate dehydrogenase kinase 1 up-regulation in activated MPs, resulting in regional pyruvate dehydrogenase inhibition. Altogether, our results demonstrate a potential for 13C MRSI of HP [1-13C]pyruvate as a neuroimaging method for assessment of inflammatory lesions. This approach could prove useful not only in MS but also in other neurological diseases presenting inflammatory components.
Keywords: hyperpolarized 13C MR spectroscopy; macrophages; metabolism; multiple sclerosis; neuroinflammation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





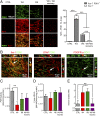
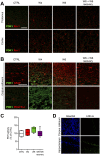
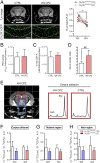
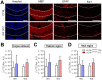
References
-
- Noseworthy JH, Lucchinetti C, Rodriguez M, Weinshenker BG. Multiple sclerosis. N Engl J Med. 2000;343:938–952. - PubMed
-
- Calabrese M, et al. Exploring the origins of grey matter damage in multiple sclerosis. Nat Rev Neurosci. 2015;16:147–158. - PubMed
-
- Quarles R, Macklin W, Morell P. Myelin formation, structure and biochemistry. In: Siegel G, Albers RW, Brady S, Price D, editors. Basic Neurochemistry: Molecular, Cellular and Medical Aspects. Elsevier; New York: 2005. pp. 51–72.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

