Effect of treatment with single total-dose intravenous iron versus daily oral iron(III)-hydroxide polymaltose on moderate puerperal iron-deficiency anemia
- PMID: 28761350
- PMCID: PMC5493201
- DOI: 10.2147/TCRM.S112227
Effect of treatment with single total-dose intravenous iron versus daily oral iron(III)-hydroxide polymaltose on moderate puerperal iron-deficiency anemia
Abstract
Background: Iron-deficiency anemia is the most common nutritional cause of anemia in pregnancy and is often responsible for puerperal anemia. Puerperal anemia can impair postpartum maternal and neonatal well-being.
Objective: To determine the effect of treatment of moderate puerperal iron-deficiency anemia using a single intravenous total-dose iron dextran versus daily single dose oral iron(III)-hydroxide polymaltose.
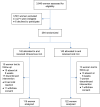
Methodology: A randomized controlled study in which postpartum women with moderate iron-deficiency anemia were randomized into treatment with either a single total-dose intravenous iron dextran or with daily single doses of oral iron(III)-hydroxide polymaltose tablets for 6 weeks. Effects on hemoglobin concentration using either method were compared at 6 weeks postpartum. Analysis was per protocol using SPSS version 17 for windows. P-values ≤0.05 were considered significant.
Results: Two hundred eighty-four women were recruited for the study: 142 women received single total dose intravenous infusion of iron dextran while 142 received daily oral iron(III)-hydroxide polymaltose tablets. Approximately 84.0% (237/282) completed the study and were analyzed including 81% (115/142) of those randomized to injectable iron therapy compared to 85.9% (122/142) of those randomized to oral treatment. The proportions of women who had attained hemoglobin concentration of at least 10 g/dL by the 6 weeks postpartum visit did not differ significantly between cases and controls (95.7% vs 94.3%; P=0.73). Similarly, the mean increases in hemoglobin following either therapeutic route were comparable (1.03±0.56 g/dL for intravenous iron and 0.97±0.46 g/dL for the oral group; P=0.42).
Conclusion: Single total-dose intravenous iron for treatment of puerperal iron-deficiency anemia was as effective as daily single doses of ferric iron tablets. For puerperal patients with iron-deficiency anemia in whom compliance with and tolerability of oral iron are not certain, a single total-dose intravenous iron can be safely offered.
Keywords: anemia; effect; intravenous; iron; puerperal.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Similar articles
-
Oral vs intravenous iron therapy for postpartum anemia: a systematic review and meta-analysis.Am J Obstet Gynecol. 2019 Jul;221(1):19-29.e3. doi: 10.1016/j.ajog.2018.12.016. Epub 2018 Dec 19. Am J Obstet Gynecol. 2019. PMID: 30578747 Free PMC article.
-
A Prospective Randomised Controlled Trial of a Single Intravenous Infusion of Ferric Carboxymaltose vs Single Intravenous Iron Polymaltose or Daily Oral Ferrous Sulphate in the Treatment of Iron Deficiency Anaemia in Pregnancy.Semin Hematol. 2018 Oct;55(4):223-234. doi: 10.1053/j.seminhematol.2018.04.006. Epub 2018 Apr 25. Semin Hematol. 2018. PMID: 30502851 Clinical Trial.
-
Addition of oral iron bisglycinate to intravenous iron sucrose for the treatment of postpartum anemia-randomized controlled trial.Am J Obstet Gynecol. 2021 Dec;225(6):668.e1-668.e9. doi: 10.1016/j.ajog.2021.06.069. Epub 2021 Jun 23. Am J Obstet Gynecol. 2021. PMID: 34171389 Clinical Trial.
-
Efficacy and safety of oral iron(III) polymaltose complex versus ferrous sulfate in pregnant women with iron-deficiency anemia: a multicenter, randomized, controlled study.J Matern Fetal Neonatal Med. 2011 Nov;24(11):1347-52. doi: 10.3109/14767058.2011.599080. Epub 2011 Aug 23. J Matern Fetal Neonatal Med. 2011. PMID: 21859366 Clinical Trial.
-
Iron(III)-hydroxide polymaltose complex in iron deficiency anemia / review and meta-analysis.Arzneimittelforschung. 2007;57(6A):431-8. doi: 10.1055/s-0031-1296692. Arzneimittelforschung. 2007. PMID: 17691593 Review.
Cited by
-
Oral vs intravenous iron therapy for postpartum anemia: a systematic review and meta-analysis.Am J Obstet Gynecol. 2019 Jul;221(1):19-29.e3. doi: 10.1016/j.ajog.2018.12.016. Epub 2018 Dec 19. Am J Obstet Gynecol. 2019. PMID: 30578747 Free PMC article.
-
Risk of Infection Associated With Administration of Intravenous Iron: A Systematic Review and Meta-analysis.JAMA Netw Open. 2021 Nov 1;4(11):e2133935. doi: 10.1001/jamanetworkopen.2021.33935. JAMA Netw Open. 2021. PMID: 34767026 Free PMC article.
-
A continuous quality improvement strategy to strengthen screening practices and facilitate the routine use of intravenous iron for treating anaemia in pregnant and postpartum women in Nigeria: a study protocol.Implement Sci Commun. 2023 Mar 7;4(1):22. doi: 10.1186/s43058-023-00400-y. Implement Sci Commun. 2023. PMID: 36882860 Free PMC article.
-
Treatment for women with postpartum iron deficiency anaemia.Cochrane Database Syst Rev. 2024 Dec 13;12(12):CD010861. doi: 10.1002/14651858.CD010861.pub3. Cochrane Database Syst Rev. 2024. PMID: 39670550
References
-
- Milasinovic L, Kapamadziga A, Dobric L, Petrovic D. Postpartum anemia–incidence and etiology. Med Pregl. 2000;53(7–8):394–399. Croatian. - PubMed
-
- Somdatta P, Reddaiah VP, Singh B. Prevalence of anemia in the postpartum period: a study of a North Indian village. Trop Doct. 2009;39(4):211–215. - PubMed
-
- Ekanem AD, Etuk SJ, Samson-Akpan U. The influence of cultural practice on puerperal anaemia. Int J Gynaecol Obstet. 1996;55(2):169–170. - PubMed
-
- Harrison KA. Maternal mortality in developing countries. Br J Obstet Gynaecol. 1989;96(1):1–3. - PubMed
-
- Brabin BJ, Hakimi M, Pelletier D. An analysis of anemia and pregnancy-related maternal mortality. J Nutr. 2001;131(2S):604S–614S. discussion 614S–615S. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources