Fungal-derived cues promote ocular autoimmunity through a Dectin-2/Card9-mediated mechanism
- PMID: 28763100
- PMCID: PMC5680064
- DOI: 10.1111/cei.13021
Fungal-derived cues promote ocular autoimmunity through a Dectin-2/Card9-mediated mechanism
Abstract
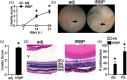
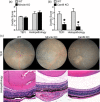
Uveitis (intraocular inflammation) is a leading cause of loss of vision. Although its aetiology is largely speculative, it is thought to arise from complex genetic-environmental interactions that break immune tolerance to generate eye-specific autoreactive T cells. Experimental autoimmune uveitis (EAU), induced by immunization with the ocular antigen, interphotoreceptor retinoid binding protein (IRBP), in combination with mycobacteria-containing complete Freund's adjuvant (CFA), has many clinical and histopathological features of human posterior uveitis. Studies in EAU have focused on defining pathogenic CD4+ T cell effector responses, such as those of T helper type 17 (Th17) cells, but the innate receptor pathways precipitating development of autoreactive, eye-specific T cells remain poorly defined. In this study, we found that fungal-derived antigens possess autoimmune uveitis-promoting function akin to CFA in conventional EAU. The capacity of commensal fungi such as Candida albicans or Saccharomyces cerevisae to promote IRBP-triggered EAU was mediated by Card9. Because Card9 is an essential signalling molecule of a subgroup of C-type lectin receptors (CLRs) important in host defence, we evaluated further the proximal Card9-activating CLRs. Using single receptor-deficient mice we identified Dectin-2, but not Mincle or Dectin-1, as a predominant mediator of fungal-promoted uveitis. Conversely, Dectin-2 activation by α-mannan reproduced the uveitic phenotype of EAU sufficiently, in a process mediated by the Card9-coupled signalling axis and interleukin (IL)-17 production. Taken together, this report relates the potential of the Dectin-2/Card9-coupled pathway in ocular autoimmunity. Not only does it contribute to understanding of how innate immune receptors orchestrate T cell-mediated autoimmunity, it also reveals a previously unappreciated ability of fungal-derived signals to promote autoimmunity.
Keywords: autoimmunity; eye; inflammation; rodent; uveitis.
© 2017 British Society for Immunology.
Figures


Similar articles
-
Mincle Activation and the Syk/Card9 Signaling Axis Are Central to the Development of Autoimmune Disease of the Eye.J Immunol. 2016 Apr 1;196(7):3148-58. doi: 10.4049/jimmunol.1502355. Epub 2016 Feb 26. J Immunol. 2016. PMID: 26921309 Free PMC article.
-
SLAT/Def6 plays a critical role in the pathogenic process of experimental autoimmune uveitis (EAU).Mol Vis. 2012;18:1858-64. Epub 2012 Jul 7. Mol Vis. 2012. PMID: 22815639 Free PMC article.
-
Murine pattern recognition receptor dectin-1 is essential in the development of experimental autoimmune uveoretinitis.Mol Immunol. 2015 Oct;67(2 Pt B):398-406. doi: 10.1016/j.molimm.2015.07.002. Epub 2015 Jul 26. Mol Immunol. 2015. PMID: 26216045
-
Autoimmunity in the immune privileged eye: pathogenic and regulatory T cells.Immunol Res. 2008;42(1-3):41-50. doi: 10.1007/s12026-008-8031-3. Immunol Res. 2008. PMID: 18629448 Free PMC article. Review.
-
Inherited CARD9 Deficiency: Invasive Disease Caused by Ascomycete Fungi in Previously Healthy Children and Adults.J Clin Immunol. 2018 Aug;38(6):656-693. doi: 10.1007/s10875-018-0539-2. Epub 2018 Aug 22. J Clin Immunol. 2018. PMID: 30136218 Free PMC article. Review.
Cited by
-
Emerging roles of Dectin-1 in noninfectious settings and in the CNS.Trends Immunol. 2021 Oct;42(10):891-903. doi: 10.1016/j.it.2021.08.005. Epub 2021 Sep 4. Trends Immunol. 2021. PMID: 34489167 Free PMC article. Review.
-
Dectin-1 limits autoimmune neuroinflammation and promotes myeloid cell-astrocyte crosstalk via Card9-independent expression of Oncostatin M.Immunity. 2021 Mar 9;54(3):484-498.e8. doi: 10.1016/j.immuni.2021.01.004. Epub 2021 Feb 12. Immunity. 2021. PMID: 33581044 Free PMC article.
-
Dectin-2-Targeted Antifungal Liposomes Exhibit Enhanced Efficacy.mSphere. 2019 Oct 30;4(5):e00715-19. doi: 10.1128/mSphere.00715-19. mSphere. 2019. PMID: 31666315 Free PMC article.
-
The Role of Fungi in the Etiology of Multiple Sclerosis.Front Neurol. 2017 Oct 16;8:535. doi: 10.3389/fneur.2017.00535. eCollection 2017. Front Neurol. 2017. PMID: 29085329 Free PMC article.
-
DectiSomes: C-type lectin receptor-targeted liposomes as pan-antifungal drugs.Adv Drug Deliv Rev. 2023 May;196:114776. doi: 10.1016/j.addr.2023.114776. Epub 2023 Mar 17. Adv Drug Deliv Rev. 2023. PMID: 36934519 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

