Frequency sensitive mechanism in low-intensity ultrasound enhanced bioeffects
- PMID: 28763448
- PMCID: PMC5538718
- DOI: 10.1371/journal.pone.0181717
Frequency sensitive mechanism in low-intensity ultrasound enhanced bioeffects
Abstract
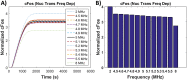
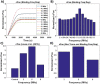
This study presents two novel theoretical models to elucidate frequency sensitive nuclear mechanisms in low-intensity ultrasound enhanced bioeffects. In contrast to the typical 1.5 MHz pulsed ultrasound regime, our group previously experimentally confirmed that ultrasound stimulation of anchored chondrocytes at resonant frequency maximized gene expression of load inducible genes which are regulatory markers for cellular response to external stimuli. However, ERK phosphorylation displayed no frequency dependency, suggesting that the biochemical mechanisms involved in enhanced gene expression is downstream of ERK phosphorylation. To elucidate such underlying mechanisms, this study presents a theoretical model of an anchored cell, representing an in vitro chondrocyte, in an ultrasound field. The model results showed that the mechanical energy storage is maximized at the chondrocyte's resonant frequency and the energy density in the nucleus is almost twice as high as in the cytoplasm. Next, a mechanochemical model was developed to link the mechanical stimulation of ultrasound and the increased mechanical energy density in the nucleus to the downstream targets of the ERK pathway. This study showed for the first time that ultrasound stimulation induces frequency dependent gene expression as a result of altered rates of transcription factors binding to chromatin.
Conflict of interest statement
Figures



References
-
- Rubin C, Bolander M, Ryaby JP, Hadjiargyrou M. The use of low-intensity ultrasound to accelerate the healing of fractures. J Bone Joint Surg Am. 2001;83: 259–270. - PubMed
-
- Martinez de Albornoz P, Khanna A, Longo UG, Forriol F, Maffulli N. The evidence of low-intensity pulsed ultrasound for in vitro, animal and human fracture healing. Br Med Bull. 2011;100: 39–57. doi: 10.1093/bmb/ldr006 - DOI - PubMed
-
- Zura R, Mehta S, Della Rocca GJ, Jones J, Steen RG. A cohort study of 4,190 patients treated with low-intensity pulsed ultrasound (LIPUS): findings in the elderly versus all patients. BMC Musculoskelet Disord. 2015;16: 45 doi: 10.1186/s12891-015-0498-1 - DOI - PMC - PubMed
-
- Xin Z, Lin G, Lei H, Lue TF, Guo Y. Clinical applications of low-inetensity pulsed ultrasound and its potential role in urology. Transl Androl Urol. 2016;5(2): 255–266. doi: 10.21037/tau.2016.02.04 - DOI - PMC - PubMed
-
- Namazi H. Effect of low-intensity pulsed ultrasound on the cartilage repair in people with mild to moderate knee osteoarthritis: a novel molecular mechanism. Arch Phys Med Rehabil. 2012;93(10): 1882 doi: 10.1016/j.apmr.2012.01.028 - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

