B Cell Requirement for Robust Regulatory T Cell Responses to Friend Retrovirus Infection
- PMID: 28765225
- PMCID: PMC5539430
- DOI: 10.1128/mBio.01122-17
B Cell Requirement for Robust Regulatory T Cell Responses to Friend Retrovirus Infection
Abstract
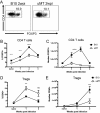
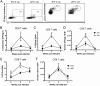
Regulatory T cells (Tregs) are immunosuppressive cells of the immune system that control autoimmune reactivity. Tregs also respond during immune reactions to infectious agents in order to limit immunopathological damage from potent effectors such as CD8+ cytolytic T lymphocytes. We have used the Friend virus (FV) model of retroviral infection in mice to investigate how viral infections induce Tregs. During acute FV infection, there is significant activation and expansion of thymus-derived (natural) Tregs that suppress virus-specific CD8+ T cell responses. Unlike conventional T cells, the responding Tregs are not virus specific, so the mechanisms that induce their expansion are of great interest. We now show that B cells provide essential signals for Treg expansion during FV infection. Treg responses are greatly diminished in B cell-deficient mice but can be restored by adoptive transfers of B cells at the time of infection. The feeble Treg responses in B cell-deficient mice are associated with enhanced virus-specific CD8+ T cell responses and accelerated virus control during the first 2 weeks of infection. In vitro experiments demonstrated that B cells promote Treg activation and proliferation through a glucocorticoid-induced receptor superfamily member 18 (GITR) ligand-dependent mechanism. Thus, B cells play paradoxically opposing roles during FV infection. They provide proliferative signals to immunsosuppressive Tregs, which slows early virus control, and they also produce virus-specific antibodies, which are essential for long-term virus control.IMPORTANCE When infectious agents invade a host, numerous immunological mechanisms are deployed to limit their replication, neutralize their spread, and destroy the host cells harboring the infection. Since immune responses also have a strong capacity to damage host cells and tissues, their magnitude, potency, and duration are under regulatory control. Regulatory T cells are an important component of this control, and the mechanisms that induce them to respond and exert immunosuppressive regulation are of great interest. In the current report, we show that B cells, the cells responsible for making pathogen-specific antibodies, are also involved in promoting the expansion of regulatory T cells during a retroviral infection. In vitro studies demonstrated that they do so via stimulation of the Tregs through interactions between cell surface molecules: GITR interactions with its ligand (GITRL) on B cells and GITR on regulatory T cells. These findings point the way toward therapeutics to better treat infections and autoimmune diseases.
Keywords: B cells; regulatory T cells; retroviruses; tumor necrosis factor receptors.
Figures




References
-
- Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M. 1995. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol 155:1151–1164. - PubMed
-
- Guilliams M, Oldenhove G, Noel W, Hérin M, Brys L, Loi P, Flamand V, Moser M, De Baetselier P, Beschin A. 2007. African trypanosomiasis: naturally occurring regulatory T cells favor trypanotolerance by limiting pathology associated with sustained type 1 inflammation. J Immunol 179:2748–2757. doi:10.4049/jimmunol.179.5.2748. - DOI - PubMed
-
- Hesse M, Piccirillo CA, Belkaid Y, Prufer J, Mentink-Kane M, Leusink M, Cheever AW, Shevach EM, Wynn TA. 2004. The pathogenesis of schistosomiasis is controlled by cooperating IL-10-producing innate effector and regulatory T cells. J Immunol 172:3157–3166. doi:10.4049/jimmunol.172.5.3157. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

