Induction of IAPP amyloid deposition and associated diabetic abnormalities by a prion-like mechanism
- PMID: 28765400
- PMCID: PMC5584114
- DOI: 10.1084/jem.20161134
Induction of IAPP amyloid deposition and associated diabetic abnormalities by a prion-like mechanism
Abstract
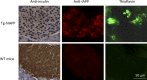
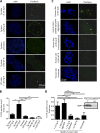
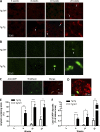
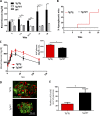
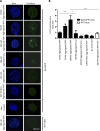
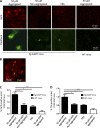
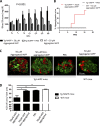
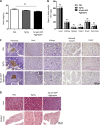
Although a large proportion of patients with type 2 diabetes (T2D) accumulate misfolded aggregates composed of the islet amyloid polypeptide (IAPP), its role in the disease is unknown. Here, we show that pancreatic IAPP aggregates can promote the misfolding and aggregation of endogenous IAPP in islet cultures obtained from transgenic mouse or healthy human pancreas. Islet homogenates immunodepleted with anti-IAPP-specific antibodies were not able to induce IAPP aggregation. Importantly, intraperitoneal inoculation of pancreatic homogenates containing IAPP aggregates into transgenic mice expressing human IAPP dramatically accelerates IAPP amyloid deposition, which was accompanied by clinical abnormalities typical of T2D, including hyperglycemia, impaired glucose tolerance, and a substantial reduction on β cell number and mass. Finally, induction of IAPP deposition and diabetic abnormalities were also induced in vivo by administration of IAPP aggregates prepared in vitro using pure, synthetic IAPP. Our findings suggest that some of the pathologic and clinical alterations of T2D might be transmissible through a similar mechanism by which prions propagate in prion diseases.
© 2017 Mukherjee et al.
Figures
Similar articles
-
Prion-Like Protein Aggregates and Type 2 Diabetes.Cold Spring Harb Perspect Med. 2017 May 1;7(5):a024315. doi: 10.1101/cshperspect.a024315. Cold Spring Harb Perspect Med. 2017. PMID: 28159831 Free PMC article. Review.
-
Molecular interaction between type 2 diabetes and Alzheimer's disease through cross-seeding of protein misfolding.Mol Psychiatry. 2017 Sep;22(9):1327-1334. doi: 10.1038/mp.2016.230. Epub 2017 Jan 3. Mol Psychiatry. 2017. PMID: 28044060 Free PMC article.
-
Spontaneous diabetes mellitus in transgenic mice expressing human islet amyloid polypeptide.Proc Natl Acad Sci U S A. 1996 Jul 9;93(14):7283-8. doi: 10.1073/pnas.93.14.7283. Proc Natl Acad Sci U S A. 1996. PMID: 8692984 Free PMC article.
-
Lipid accelerating the fibril of islet amyloid polypeptide aggravated the pancreatic islet injury in vitro and in vivo.Lipids Health Dis. 2018 Mar 9;17(1):42. doi: 10.1186/s12944-018-0694-8. Lipids Health Dis. 2018. PMID: 29523142 Free PMC article.
-
[Construction of transgenic mouse system expressing human islet amyloid polypeptide (IAPP)/amylin].Nihon Rinsho. 1994 Oct;52(10):2746-50. Nihon Rinsho. 1994. PMID: 7983808 Review. Japanese.
Cited by
-
Could α-Synuclein Modulation of Insulin and Dopamine Identify a Novel Link Between Parkinson's Disease and Diabetes as Well as Potential Therapies?Front Mol Neurosci. 2018 Dec 21;11:465. doi: 10.3389/fnmol.2018.00465. eCollection 2018. Front Mol Neurosci. 2018. PMID: 30622456 Free PMC article. Review.
-
Secretory Autophagy and Its Relevance in Metabolic and Degenerative Disease.Front Endocrinol (Lausanne). 2020 May 5;11:266. doi: 10.3389/fendo.2020.00266. eCollection 2020. Front Endocrinol (Lausanne). 2020. PMID: 32477265 Free PMC article. Review.
-
Cynomolgus Monkeys With Spontaneous Type-2-Diabetes-Mellitus-Like Pathology Develop Alpha-Synuclein Alterations Reminiscent of Prodromal Parkinson's Disease and Related Diseases.Front Neurosci. 2020 Feb 7;14:63. doi: 10.3389/fnins.2020.00063. eCollection 2020. Front Neurosci. 2020. PMID: 32116510 Free PMC article.
-
Pathogenic Carboxyl Ester Lipase (CEL) Variants Interact with the Normal CEL Protein in Pancreatic Cells.Cells. 2020 Jan 18;9(1):244. doi: 10.3390/cells9010244. Cells. 2020. PMID: 31963687 Free PMC article.
-
Diabetes mellitus and Parkinson's disease: dangerous liaisons between insulin and dopamine.Neural Regen Res. 2022 Mar;17(3):523-533. doi: 10.4103/1673-5374.320965. Neural Regen Res. 2022. PMID: 34380882 Free PMC article. Review.
References
-
- Betsholtz C., Svensson V., Rorsman F., Engström U., Westermark G.T., Wilander E., Johnson K., and Westermark P.. 1989. Islet amyloid polypeptide (IAPP): cDNA cloning and identification of an amyloidogenic region associated with the species-specific occurrence of age-related diabetes mellitus. Exp. Cell Res. 183:484–493. 10.1016/0014-4827(89)90407-2 - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

