Silicon-Tethered Strategies for C-H Functionalization Reactions
- PMID: 28771325
- PMCID: PMC5724575
- DOI: 10.1021/acs.accounts.7b00306
Silicon-Tethered Strategies for C-H Functionalization Reactions
Abstract
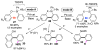
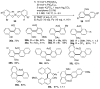
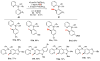
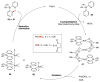
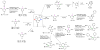
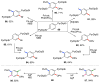
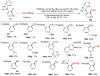
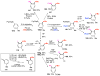
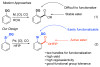
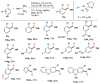
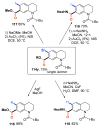
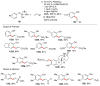
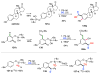
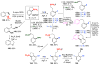
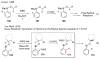
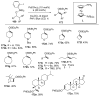
Selective and efficient functionalization of ubiquitous C-H bonds is the Holy Grail of organic synthesis. Most advances in this area rely on employment of strongly or weakly coordinating directing groups (DGs) which have proven effective for transition-metal-catalyzed functionalization of C(sp2)-H and C(sp3)-H bonds. Although most directing groups are important functionalities in their own right, in certain cases, the DGs become static entities that possess very little synthetic leverage. Moreover, some of the DGs employed are cumbersome or unpractical to remove, which precludes the use of this approach in synthesis. It is believed, that development of a set of easily installable and removable/modifiable DGs for C-H functionalization would add tremendous value to the growing area of directed functionalization, and hence would promote its use in synthesis and late-stage functionalization of complex molecules. In particular, silicon tethers have long provided leverage in organic synthesis as easily installable and removable/modifiable auxiliaries for a variety of processes, including radical transformations, cycloaddition reactions, and a number of TM-catalyzed methods, including ring-closing metathesis (RCM) and cross-coupling reactions. Employment of Si-tethers is highly attractive for several reasons: (1) they are easy to handle/synthesize and are relatively stable; (2) they utilize cheap and abundant silicon precursors; and (3) Si-tethers are easily installable and removable/modifiable. Hence, development of Si-tethers for C-H functionalization reactions is appealing not only from a practical but also from a synthetic standpoint, since the Si-tether can provide an additional handle for diversification of organic molecules post-C-H functionalization. Over the past few years, we developed a set of Si-tether approaches for C-H functionalization reactions. The developed Si-tethers can be categorized into four types: (Type-1) Si-tethers possessing a reacting group, where the reacting group is delivered to the site of functionalization; (Type-2) Si-tethers possessing a DG, designed for selective C(sp2)-H functionalization of arenes; (Type-3) reactive Si-tethers for C-H silylation of organic molecules; and finally, (Type-4) reactive Si-tethers containing a DG, developed for selective C-H silylation/hydroxylation of challenging C(sp3)-H bonds. In this Account, we outline our advances on the employment of silicon auxiliaries for directed C-H functionalization reactions. The discussion of the strategies for employment of different Si-tethers, functionalization/modification of silicon tethers, and the methodological developments on C-C, C-X, C-O, and C-Si bond forming reactions via silicon tethers will also be presented. While the work described herein presents a substantial advance for the area of C-H functionalization, challenges still remain. The use of noble metals are required for the C-H functionalization methods presented herein. Also, the need for stoichiometric use of high molecular weight silicon auxiliaries is a shortcoming of the presented concept.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



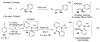


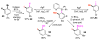





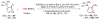
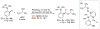








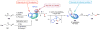


References
-
- Zhang F, Spring DR, Arene CH. Functionalization Using a Removable/modifiable or a Traceless Directing Group Strategy. Chem Soc Rev. 2014;43:6906–6919. - PubMed
-
- Bols M, Skrydstrup T. Silicon-Tethered Reactions. Chem Rev. 1995;95:1253–1277.
- Bracegirdle S, Anderson EA. Recent Advances in the Use of Temporary Silicon Tethers in Metal-mediated Reactions. Chem Soc Rev. 2010;39:4114–4129. - PubMed
-
- Hassan J, Sévignon M, Gozzi C, Schulz E, Lemaire M. Aryl–Aryl Bond Formation One Century after the Discovery of the Ullmann Reaction. Chem Rev. 2002;102:1359–1470. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

