Laser Sintered Magnesium-Calcium Silicate/Poly-ε-Caprolactone Scaffold for Bone Tissue Engineering
- PMID: 28772425
- PMCID: PMC5344575
- DOI: 10.3390/ma10010065
Laser Sintered Magnesium-Calcium Silicate/Poly-ε-Caprolactone Scaffold for Bone Tissue Engineering
Abstract
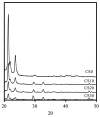
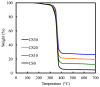
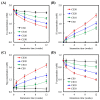
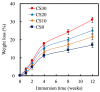
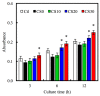
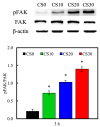
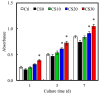
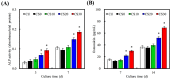
In this study, we manufacture and analyze bioactive magnesium-calcium silicate/poly-ε-caprolactone (Mg-CS/PCL) 3D scaffolds for bone tissue engineering. Mg-CS powder was incorporated into PCL, and we fabricated the 3D scaffolds using laser sintering technology. These scaffolds had high porosity and interconnected-design macropores and structures. As compared to pure PCL scaffolds without an Mg-CS powder, the hydrophilic properties and degradation rate are also improved. For scaffolds with more than 20% Mg-CS content, the specimens become completely covered by a dense bone-like apatite layer after soaking in simulated body fluid for 1 day. In vitro analyses were directed using human mesenchymal stem cells (hMSCs) on all scaffolds that were shown to be biocompatible and supported cell adhesion and proliferation. Increased focal adhesion kinase and promoted cell adhesion behavior were observed after an increase in Mg-CS content. In addition, the results indicate that the Mg-CS quantity in the composite is higher than 10%, and the quantity of cells and osteogenesis-related protein of hMSCs is stimulated by the Si ions released from the Mg-CS/PCL scaffolds when compared to PCL scaffolds. Our results proved that 3D Mg-CS/PCL scaffolds with such a specific ionic release and good degradability possessed the ability to promote osteogenetic differentiation of hMSCs, indicating that they might be promising biomaterials with potential for next-generation bone tissue engineering scaffolds.
Keywords: calcium silicate; human marrow stem cells; laser sintering; osteogenesis; scaffold.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Bioactive calcium silicate/poly-ε-caprolactone composite scaffolds 3D printed under mild conditions for bone tissue engineering.J Mater Sci Mater Med. 2017 Dec 27;29(1):11. doi: 10.1007/s10856-017-6020-6. J Mater Sci Mater Med. 2017. PMID: 29282550
-
Tissue engineering scaffolds of mesoporous magnesium silicate and poly(ε-caprolactone)-poly(ethylene glycol)-poly(ε-caprolactone) composite.J Mater Sci Mater Med. 2014 Jun;25(6):1415-24. doi: 10.1007/s10856-014-5183-7. Epub 2014 Mar 5. J Mater Sci Mater Med. 2014. PMID: 24595904
-
Selective laser sintering fabrication of nano-hydroxyapatite/poly-ε-caprolactone scaffolds for bone tissue engineering applications.Int J Nanomedicine. 2013;8:4197-213. doi: 10.2147/IJN.S50685. Epub 2013 Nov 1. Int J Nanomedicine. 2013. PMID: 24204147 Free PMC article.
-
Mesoporous magnesium silicate-incorporated poly(ε-caprolactone)-poly(ethylene glycol)-poly(ε-caprolactone) bioactive composite beneficial to osteoblast behaviors.Int J Nanomedicine. 2014 May 27;9:2665-75. doi: 10.2147/IJN.S59040. eCollection 2014. Int J Nanomedicine. 2014. PMID: 24920903 Free PMC article.
-
Effect of Strontium Substitution on the Physicochemical Properties and Bone Regeneration Potential of 3D Printed Calcium Silicate Scaffolds.Int J Mol Sci. 2019 Jun 3;20(11):2729. doi: 10.3390/ijms20112729. Int J Mol Sci. 2019. PMID: 31163656 Free PMC article.
Cited by
-
3D Printing-Encompassing the Facets of Dentistry.Front Bioeng Biotechnol. 2018 Nov 22;6:172. doi: 10.3389/fbioe.2018.00172. eCollection 2018. Front Bioeng Biotechnol. 2018. PMID: 30525032 Free PMC article. Review.
-
Therapeutic Metallic Ions in Bone Tissue Engineering: A Systematic Review of The Literature.Iran J Pharm Res. 2019 Fall;18(Suppl1):101-118. doi: 10.22037/ijpr.2020.112641.13894. Iran J Pharm Res. 2019. PMID: 32802092 Free PMC article. Review.
-
Bioabsorbable Composites Based on Polymeric Matrix (PLA and PCL) Reinforced with Magnesium (Mg) for Use in Bone Regeneration Therapy: Physicochemical Properties and Biological Evaluation.Polymers (Basel). 2023 Dec 11;15(24):4667. doi: 10.3390/polym15244667. Polymers (Basel). 2023. PMID: 38139919 Free PMC article. Review.
-
Osteogenic Differentiation Potential of Adipose-Derived Mesenchymal Stem Cells Cultured on Magnesium Oxide/Polycaprolactone Nanofibrous Scaffolds for Improving Bone Tissue Reconstruction.Adv Pharm Bull. 2022 Jan;12(1):142-154. doi: 10.34172/apb.2022.015. Epub 2020 Sep 22. Adv Pharm Bull. 2022. PMID: 35517875 Free PMC article.
-
PCL-Based Composite Scaffold Matrices for Tissue Engineering Applications.Mol Biotechnol. 2018 Jul;60(7):506-532. doi: 10.1007/s12033-018-0084-5. Mol Biotechnol. 2018. PMID: 29761314 Review.
References
-
- Su C.C., Kao C.T., Hung C.J., Chen Y.J., Huang T.H., Shie M.Y. Regulation of physicochemical properties, osteogenesis activity, and fibroblast growth factor-2 release ability of β-tricalcium phosphate for bone cement by calcium silicate. Mater. Sci. Eng. C Mater. Biol. Appl. 2014;37:156–163. doi: 10.1016/j.msec.2014.01.010. - DOI - PubMed
-
- Qi X., Huang Y., Han D., Zhang J., Cao J., Jin X., Huang J., Li X., Wang T.M. Three-dimensional poly (ε-caprolactone)/hydroxyapatite/collagen scaffolds incorporating bone marrow mesenchymal stem cells for the repair of bone defects. Biomed. Mater. 2016;11:025005. doi: 10.1088/1748-6041/11/2/025005. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

