Evaluation of 3D-Printed Polycaprolactone Scaffolds Coated with Freeze-Dried Platelet-Rich Plasma for Bone Regeneration
- PMID: 28773189
- PMCID: PMC5551874
- DOI: 10.3390/ma10070831
Evaluation of 3D-Printed Polycaprolactone Scaffolds Coated with Freeze-Dried Platelet-Rich Plasma for Bone Regeneration
Abstract
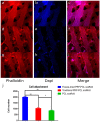
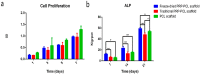
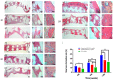
Three-dimensional printing is one of the most promising techniques for the manufacturing of scaffolds for bone tissue engineering. However, a pure scaffold is limited by its biological properties. Platelet-rich plasma (PRP) has been shown to have the potential to improve the osteogenic effect. In this study, we improved the biological properties of scaffolds by coating 3D-printed polycaprolactone (PCL) scaffolds with freeze-dried and traditionally prepared PRP, and we evaluated these scaffolds through in vitro and in vivo experiments. In vitro, we evaluated the interaction between dental pulp stem cells (DPSCs) and the scaffolds by measuring cell proliferation, alkaline phosphatase (ALP) activity, and osteogenic differentiation. The results showed that freeze-dried PRP significantly enhanced ALP activity and the mRNA expression levels of osteogenic genes (ALP, RUNX2 (runt-related gene-2), OCN (osteocalcin), OPN (osteopontin)) of DPSCs (p < 0.05). In vivo, 5 mm calvarial defects were created, and the PRP-PCL scaffolds were implanted. The data showed that compared with traditional PRP-PCL scaffolds or bare PCL scaffolds, the freeze-dried PRP-PCL scaffolds induced significantly greater bone formation (p < 0.05). All these data suggest that coating 3D-printed PCL scaffolds with freeze-dried PRP can promote greater osteogenic differentiation of DPSCs and induce more bone formation, which may have great potential in future clinical applications.
Keywords: 3D-printed scaffold; bone regeneration; platelet-rich plasma; polycaprolactone.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




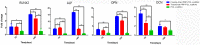

Similar articles
-
Nano-hydroxy apatite/chitosan/gelatin scaffolds enriched by a combination of platelet-rich plasma and fibrin glue enhance proliferation and differentiation of seeded human dental pulp stem cells.Biomed Pharmacother. 2019 Jan;109:1924-1931. doi: 10.1016/j.biopha.2018.11.072. Epub 2018 Nov 26. Biomed Pharmacother. 2019. PMID: 30551447
-
Surface modification of 3D-printed porous scaffolds via mussel-inspired polydopamine and effective immobilization of rhBMP-2 to promote osteogenic differentiation for bone tissue engineering.Acta Biomater. 2016 Aug;40:182-191. doi: 10.1016/j.actbio.2016.02.006. Epub 2016 Feb 8. Acta Biomater. 2016. PMID: 26868173
-
Dental pulp-derived stromal cells exhibit a higher osteogenic potency than bone marrow-derived stromal cells in vitro and in a porcine critical-size bone defect model.SICOT J. 2016 Apr 20;2:16. doi: 10.1051/sicotj/2016004. SICOT J. 2016. PMID: 27163105 Free PMC article.
-
The Application of Polycaprolactone in Three-Dimensional Printing Scaffolds for Bone Tissue Engineering.Polymers (Basel). 2021 Aug 17;13(16):2754. doi: 10.3390/polym13162754. Polymers (Basel). 2021. PMID: 34451293 Free PMC article. Review.
-
Polycaprolactone for Hard Tissue Regeneration: Scaffold Design and In Vivo Implications.Bioengineering (Basel). 2025 Jan 8;12(1):46. doi: 10.3390/bioengineering12010046. Bioengineering (Basel). 2025. PMID: 39851320 Free PMC article. Review.
Cited by
-
The Applications of 3D Printing for Craniofacial Tissue Engineering.Micromachines (Basel). 2019 Jul 17;10(7):480. doi: 10.3390/mi10070480. Micromachines (Basel). 2019. PMID: 31319522 Free PMC article. Review.
-
Immobilization and Release of Platelet-Rich Plasma from Modified Nanofibers Studied by Advanced X-ray Photoelectron Spectroscopy Analyses.Polymers (Basel). 2023 Mar 14;15(6):1440. doi: 10.3390/polym15061440. Polymers (Basel). 2023. PMID: 36987220 Free PMC article.
-
In Vivo Bone Regeneration Capacity of Multiscale Porous Polycaprolactone-Based High Internal Phase Emulsion (PolyHIPE) Scaffolds in a Rat Calvarial Defect Model.ACS Appl Mater Interfaces. 2023 Jun 14;15(23):27696-27705. doi: 10.1021/acsami.3c04362. Epub 2023 May 30. ACS Appl Mater Interfaces. 2023. PMID: 37253168 Free PMC article.
-
Fabrication of Mechanically Enhanced, Suturable, Fibrous Hydrogel Membranes.Membranes (Basel). 2023 Jan 16;13(1):116. doi: 10.3390/membranes13010116. Membranes (Basel). 2023. PMID: 36676923 Free PMC article.
-
3D Printed and Bioprinted Membranes and Scaffolds for the Periodontal Tissue Regeneration: A Narrative Review.Membranes (Basel). 2022 Sep 19;12(9):902. doi: 10.3390/membranes12090902. Membranes (Basel). 2022. PMID: 36135920 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

