Crystal Structures and Mechanical Properties of Ca₂C at High Pressure
- PMID: 28773695
- PMCID: PMC5456912
- DOI: 10.3390/ma9070570
Crystal Structures and Mechanical Properties of Ca₂C at High Pressure
Abstract
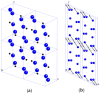
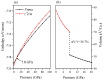
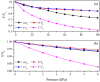
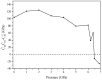
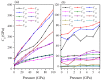
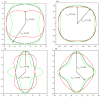
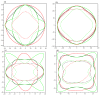
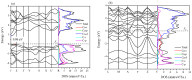
Recently, a new high-pressure semiconductor phase of Ca₂C (space group Pnma) was successfully synthesized, it has a low-pressure metallic phase (space group C2/m). In this paper, a systematic investigation of the pressure-induced phase transition of Ca₂C is studied on the basis of first-principles calculations. The calculated enthalpy reveals that the phase transition which transforms from C2/m-Ca₂C to Pnma-Ca₂C occurs at 7.8 GPa, and it is a first-order phase transition with a volume drop of 26.7%. The calculated elastic constants show that C2/m-Ca₂C is mechanically unstable above 6.4 GPa, indicating that the structural phase transition is due to mechanical instability. Both of the two phases exhibit the elastic anisotropy. The semiconductivity of Pnma-Ca₂C and the metallicity of C2/m-Ca₂C have been demonstrated by the electronic band structure calculations. The quasi-direct band gap of Pnma-Ca₂C at 0 GPa is 0.86 eV. Furthermore, the detailed analysis of the total and partial density of states is performed to show the specific contribution to the Fermi level.
Keywords: Ca2C; first-principles calculations; pressure-induced phase transition.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Pnma-BN: Another Boron Nitride Polymorph with Interesting Physical Properties.Nanomaterials (Basel). 2016 Dec 28;7(1):3. doi: 10.3390/nano7010003. Nanomaterials (Basel). 2016. PMID: 28336837 Free PMC article.
-
Pressure effects on the structures and electronic properties of halide perovskite CsPbX3 (X = I, Br, Cl).Phys Chem Chem Phys. 2021 Feb 7;23(5):3479-3484. doi: 10.1039/d0cp05892k. Epub 2021 Jan 28. Phys Chem Chem Phys. 2021. PMID: 33507187
-
Electronic and elastic properties of CaF(2) under high pressure from ab initio calculations.J Phys Condens Matter. 2009 Oct 14;21(41):415501. doi: 10.1088/0953-8984/21/41/415501. Epub 2009 Sep 23. J Phys Condens Matter. 2009. PMID: 21693988
-
Structural, mechanical and electronic properties and hardness of ionic vanadium dihydrides under pressure from first-principles computations.Sci Rep. 2020 Jun 1;10(1):8868. doi: 10.1038/s41598-020-65910-4. Sci Rep. 2020. PMID: 32483252 Free PMC article.
-
First-principles study of pressure-induced phase transitions, mechanical and thermodynamic properties of ThBC.J Phys Condens Matter. 2021 Nov 4;34(4). doi: 10.1088/1361-648X/ac3103. J Phys Condens Matter. 2021. PMID: 34666326
Cited by
-
Mechanical and Electronic Properties of XC₆ and XC12.Materials (Basel). 2016 Aug 25;9(9):726. doi: 10.3390/ma9090726. Materials (Basel). 2016. PMID: 28773847 Free PMC article.
References
-
- Fan Q., Chai C., Wei Q., Yang Y., Yang Q., Chen P., Xing M., Zhang J., Yao R. Prediction of novel phase of silicon and Si-Ge alloys. J. Solid State Chem. 2016;233:471–483. doi: 10.1016/j.jssc.2015.11.021. - DOI
-
- Wei Q., Yan H., Zhu X., Lin Z., Yao R. Theoretical investigations on the elastic and thermodynamic properties of Rhenium Phosphide. Z. Naturforsch. A. 2016;71:1–8. doi: 10.1515/zna-2014-0301. - DOI
-
- Gauzzi A., Takashima S., Takeshita N., Terakura C., Takagi H., Emery N., Hérold C., Lagrange P., Loupias G. Enhancement of superconductivity and evidence of structural instability in intercalated Graphite CaC6 under high pressure. Phys. Rev. Lett. 2007;98:067002. doi: 10.1103/PhysRevLett.98.067002. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

