Transglutaminase 2 in pulmonary and cardiac tissue remodeling in experimental pulmonary hypertension
- PMID: 28775095
- PMCID: PMC5792178
- DOI: 10.1152/ajplung.00170.2017
Transglutaminase 2 in pulmonary and cardiac tissue remodeling in experimental pulmonary hypertension
Abstract
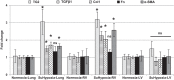
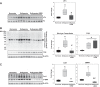
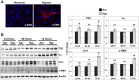
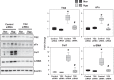
Tissue matrix remodeling and fibrosis leading to loss of pulmonary arterial and right ventricular compliance are important features of both experimental and clinical pulmonary hypertension (PH). We have previously reported that transglutaminase 2 (TG2) is involved in PH development while others have shown it to be a cross-linking enzyme that participates in remodeling of extracellular matrix in fibrotic diseases in general. In the present studies, we used a mouse model of experimental PH (Sugen 5416 and hypoxia; SuHypoxia) and cultured primary human cardiac and pulmonary artery adventitial fibroblasts to evaluate the relationship of TG2 to the processes of fibrosis, protein cross-linking, extracellular matrix collagen accumulation, and fibroblast-to-myofibroblast transformation. We report here that TG2 expression and activity as measured by serotonylated fibronectin and protein cross-linking activity along with fibrogenic markers are significantly elevated in lungs and right ventricles of SuHypoxic mice with PH. Similarly, TG2 expression and activity, protein cross-linking activity, and fibrogenic markers are significantly increased in cultured cardiac and pulmonary artery adventitial fibroblasts in response to hypoxia exposure. Pharmacological inhibition of TG2 activity with ERW1041E significantly reduced hypoxia-induced cross-linking activity and synthesis of collagen 1 and α-smooth muscle actin in both the in vivo and in vitro studies. TG2 short interfering RNA had a similar effect in vitro. Our results suggest that TG2 plays an important role in hypoxia-induced pulmonary and right ventricular tissue matrix remodeling in the development of PH.
Keywords: pulmonary hypertension; pulmonary remodeling; right ventricular remodeling; tissue fibrosis; transglutaminase 2.
Copyright © 2017 the American Physiological Society.
Figures



Similar articles
-
Lung-specific interleukin 6 mediated transglutaminase 2 activation and cardiopulmonary fibrogenesis.Front Immunol. 2024 Apr 8;15:1371706. doi: 10.3389/fimmu.2024.1371706. eCollection 2024. Front Immunol. 2024. PMID: 38650935 Free PMC article.
-
Glycolysis regulated transglutaminase 2 activation in cardiopulmonary fibrogenic remodeling.FASEB J. 2020 Jan;34(1):930-944. doi: 10.1096/fj.201902155R. Epub 2019 Nov 28. FASEB J. 2020. PMID: 31914588 Free PMC article.
-
Role of hypoxia-induced transglutaminase 2 in pulmonary artery smooth muscle cell proliferation.Am J Physiol Lung Cell Mol Physiol. 2014 Oct 1;307(7):L576-85. doi: 10.1152/ajplung.00162.2014. Epub 2014 Aug 15. Am J Physiol Lung Cell Mol Physiol. 2014. PMID: 25128524 Free PMC article.
-
Transglutaminase 2-mediated serotonylation in pulmonary hypertension.Am J Physiol Lung Cell Mol Physiol. 2014 Feb 15;306(4):L309-15. doi: 10.1152/ajplung.00321.2013. Epub 2013 Dec 27. Am J Physiol Lung Cell Mol Physiol. 2014. PMID: 24375797 Free PMC article. Review.
-
Role of extracellular matrix in the pathogenesis of pulmonary arterial hypertension.Am J Physiol Heart Circ Physiol. 2018 Nov 1;315(5):H1322-H1331. doi: 10.1152/ajpheart.00136.2018. Epub 2018 Aug 24. Am J Physiol Heart Circ Physiol. 2018. PMID: 30141981 Free PMC article. Review.
Cited by
-
Lung-specific interleukin 6 mediated transglutaminase 2 activation and cardiopulmonary fibrogenesis.Front Immunol. 2024 Apr 8;15:1371706. doi: 10.3389/fimmu.2024.1371706. eCollection 2024. Front Immunol. 2024. PMID: 38650935 Free PMC article.
-
Increased Transglutaminase 2 Expression and Activity in Rodent Models of Obesity/Metabolic Syndrome and Aging.Front Physiol. 2020 Sep 15;11:560019. doi: 10.3389/fphys.2020.560019. eCollection 2020. Front Physiol. 2020. PMID: 33041859 Free PMC article.
-
Mechanobiological Feedback in Pulmonary Vascular Disease.Front Physiol. 2018 Jul 25;9:951. doi: 10.3389/fphys.2018.00951. eCollection 2018. Front Physiol. 2018. PMID: 30090065 Free PMC article. Review.
-
CrossTalk opposing view: The mouse SuHx model is not a good model of pulmonary arterial hypertension.J Physiol. 2019 Feb;597(4):979-981. doi: 10.1113/JP275865. Epub 2018 Nov 29. J Physiol. 2019. PMID: 30499185 Free PMC article. No abstract available.
-
Cystamine reduces vascular stiffness in Western diet-fed female mice.Am J Physiol Heart Circ Physiol. 2022 Feb 1;322(2):H167-H180. doi: 10.1152/ajpheart.00431.2021. Epub 2021 Dec 10. Am J Physiol Heart Circ Physiol. 2022. PMID: 34890280 Free PMC article.
References
-
- Alaa M, Abdellatif M, Tavares-Silva M, Oliveira-Pinto J, Lopes L, Leite S, Leite-Moreira AF, Lourenço AP. Right ventricular end-diastolic stiffness heralds right ventricular failure in monocrotaline-induced pulmonary hypertension. Am J Physiol Heart Circ Physiol 311: H1004–H1013, 2016. doi:10.1152/ajpheart.00202.2016. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

