1,6-Cyclophellitol Cyclosulfates: A New Class of Irreversible Glycosidase Inhibitor
- PMID: 28776021
- PMCID: PMC5532717
- DOI: 10.1021/acscentsci.7b00214
1,6-Cyclophellitol Cyclosulfates: A New Class of Irreversible Glycosidase Inhibitor
Abstract
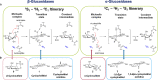
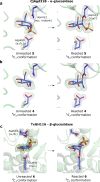
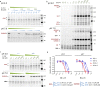
The essential biological roles played by glycosidases, coupled to the diverse therapeutic benefits of pharmacologically targeting these enzymes, provide considerable motivation for the development of new inhibitor classes. Cyclophellitol epoxides and aziridines are recently established covalent glycosidase inactivators. Inspired by the application of cyclic sulfates as electrophilic equivalents of epoxides in organic synthesis, we sought to test whether cyclophellitol cyclosulfates would similarly act as irreversible glycosidase inhibitors. Here we present the synthesis, conformational analysis, and application of novel 1,6-cyclophellitol cyclosulfates. We show that 1,6-epi-cyclophellitol cyclosulfate (α-cyclosulfate) is a rapidly reacting α-glucosidase inhibitor whose 4C1 chair conformation matches that adopted by α-glucosidase Michaelis complexes. The 1,6-cyclophellitol cyclosulfate (β-cyclosulfate) reacts more slowly, likely reflecting its conformational restrictions. Selective glycosidase inhibitors are invaluable as mechanistic probes and therapeutic agents, and we propose cyclophellitol cyclosulfates as a valuable new class of carbohydrate mimetics for application in these directions.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

