DACH1 stimulates shear stress-guided endothelial cell migration and coronary artery growth through the CXCL12-CXCR4 signaling axis
- PMID: 28779009
- PMCID: PMC5580653
- DOI: 10.1101/gad.301549.117
DACH1 stimulates shear stress-guided endothelial cell migration and coronary artery growth through the CXCL12-CXCR4 signaling axis
Abstract
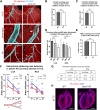
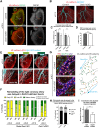
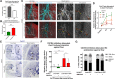
Sufficient blood flow to tissues relies on arterial blood vessels, but the mechanisms regulating their development are poorly understood. Many arteries, including coronary arteries of the heart, form through remodeling of an immature vascular plexus in a process triggered and shaped by blood flow. However, little is known about how cues from fluid shear stress are translated into responses that pattern artery development. Here, we show that mice lacking endothelial Dach1 had small coronary arteries, decreased endothelial cell polarization, and reduced expression of the chemokine Cxcl12 Under shear stress in culture, Dach1 overexpression stimulated endothelial cell polarization and migration against flow, which was reversed upon CXCL12/CXCR4 inhibition. In vivo, DACH1 was expressed during early arteriogenesis but was down in mature arteries. Mature artery-type shear stress (high, uniform laminar) specifically down-regulated DACH1, while the remodeling artery-type flow (low, variable) maintained DACH1 expression. Together, our data support a model in which DACH1 stimulates coronary artery growth by activating Cxcl12 expression and endothelial cell migration against blood flow into developing arteries. This activity is suppressed once arteries reach a mature morphology and acquire high, laminar flow that down-regulates DACH1. Thus, we identified a mechanism by which blood flow quality balances artery growth and maturation.
Keywords: arteriogenesis; cell migration; coronary artery development; endothelial cell biology; mechanotransduction; vascular biology.
© 2017 Chang et al.; Published by Cold Spring Harbor Laboratory Press.
Figures




References
-
- Ara T, Tokoyoda K, Okamoto R, Koni PA, Nagasawa T. 2005. The role of CXCL12 in the organ-specific process of artery formation. Blood 105: 3155–3161. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
