Role of depth of response and MTHFR genotype as predictors of fluorouracil rechallenge therapy for refractory metastatic colorectal cancer
- PMID: 28781687
- PMCID: PMC5530182
- DOI: 10.3892/ol.2017.6414
Role of depth of response and MTHFR genotype as predictors of fluorouracil rechallenge therapy for refractory metastatic colorectal cancer
Abstract
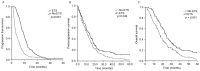
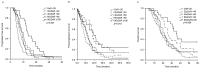
There is limited data on the clinical and biological parameters that enable the prediction of the benefits derived from additional chemotherapy after disease progression compared with standard chemotherapy in patients with metastatic colorectal cancer (mCRC). The present study evaluated the role of tumor response as a clinical parameter and single nucleotide polymorphisms (SNPs) as a biomarker to predict the benefit of additional 5-fluorouracil (5-FU) rechallenge chemotherapy in patients with refractory mCRC. Tumor responses were retrospectively reviewed based on the Response Evaluation Criteria in Solid Tumors, early tumor shrinkage (ETS) and depth of response (DoR) following first-line chemotherapy in patients with stage IV CRC. Together with these parameters, SNPs known to be associated with the response to chemotherapy were analyzed with survival outcomes. In total, the tumor responses of 242 patients with mCRC were evaluated. Overall response and ETS were identified in 110 (45.4%) and 103 patients (42.6%), respectively, and the median DoR was 38.5±30.08%. ETS and DoR were significantly associated with survival outcomes, including progression-free survival, post-progression survival and overall survival. Among these patients, SNPs were analyzed in 171 patients. X-ray repair cross complementing 1 (XRCC1) (AG/AA) with a DoR >60%, good performance status and the absence of bone lesions were associated with improved overall survival. In patients receiving third-line chemotherapy with 5-FU rechallenge therapy, the methylenetretrahydrofolate reductase (MTHFR) (C677T) CC genotype and a DoR >60% were significantly associated with a good prognosis in multivariate analysis. XRCC1 (AG/AA) was also associated with a good prognosis in patients with mCRC. Patients with a DoR >60% following first-line chemotherapy and a MTHFR (C677T) CC genotype exhibited a survival benefit from 5-FU retreatment. Therefore, the DoR and MTHFR genotype are potential markers for selecting patients with refractory mCRC that would benefit from 5-FU rechallenge therapy.
Keywords: chemotherapy; colorectal cancer; depth of response; early tumor shrinkage; polymorphism.
Figures
Similar articles
-
A narrative review: depth of response as a predictor of the long-term outcomes for solid tumors.Transl Cancer Res. 2021 Feb;10(2):1119-1130. doi: 10.21037/tcr-20-2547. Transl Cancer Res. 2021. PMID: 35116438 Free PMC article. Review.
-
Polymorphisms of MTHFR C677T and A1298C associated with survival in patients with colorectal cancer treated with 5-fluorouracil-based chemotherapy.Int J Clin Oncol. 2017 Jun;22(3):484-493. doi: 10.1007/s10147-016-1080-z. Epub 2017 Jan 2. Int J Clin Oncol. 2017. PMID: 28044213 Clinical Trial.
-
Early Tumor Shrinkage and Depth of Response Evaluation in Metastatic Pancreatic Cancer Treated with First Line Chemotherapy: An Observational Retrospective Cohort Study.Cancers (Basel). 2019 Jul 4;11(7):939. doi: 10.3390/cancers11070939. Cancers (Basel). 2019. PMID: 31277449 Free PMC article.
-
Which gene is a dominant predictor of response during FOLFOX chemotherapy for the treatment of metastatic colorectal cancer, the MTHFR or XRCC1 gene?Ann Surg Oncol. 2006 Nov;13(11):1379-85. doi: 10.1245/s10434-006-9112-y. Epub 2006 Sep 29. Ann Surg Oncol. 2006. PMID: 17009149
-
Early tumour shrinkage (ETS) and depth of response (DpR) in the treatment of patients with metastatic colorectal cancer (mCRC).Eur J Cancer. 2015 Sep;51(14):1927-36. doi: 10.1016/j.ejca.2015.06.116. Epub 2015 Jul 15. Eur J Cancer. 2015. PMID: 26188850 Review.
Cited by
-
Recent Updates on Mechanisms of Resistance to 5-Fluorouracil and Reversal Strategies in Colon Cancer Treatment.Biology (Basel). 2021 Aug 31;10(9):854. doi: 10.3390/biology10090854. Biology (Basel). 2021. PMID: 34571731 Free PMC article. Review.
-
SNPs in predicting clinical efficacy and toxicity of chemotherapy: walking through the quicksand.Oncotarget. 2018 May 18;9(38):25355-25382. doi: 10.18632/oncotarget.25256. eCollection 2018 May 18. Oncotarget. 2018. PMID: 29861877 Free PMC article. Review.
-
A narrative review: depth of response as a predictor of the long-term outcomes for solid tumors.Transl Cancer Res. 2021 Feb;10(2):1119-1130. doi: 10.21037/tcr-20-2547. Transl Cancer Res. 2021. PMID: 35116438 Free PMC article. Review.
References
-
- Van Cutsem E, Köhne CH, Láng I, Folprecht G, Nowacki MP, Cascinu S, Shchepotin I, Maurel J, Cunningham D, Tejpar S, et al. Cetuximab plus irinotecan, fluorouracil, and leucovorin as first-line treatment for metastatic colorectal cancer: Updated analysis of overall survival according to tumor KRAS and BRAF mutation status. J Clin Oncol. 2011;29:2011–2019. doi: 10.1200/JCO.2010.33.5091. - DOI - PubMed
-
- Grothey A, Van Cutsem E, Sobrero A, Siena S, Falcone A, Ychou M, Humblet Y, Bouché O, Mineur L, Barone C, et al. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): An international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381:303–312. doi: 10.1016/S0140-6736(12)61900-X. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
