Histone deacetylase 1 and 2 are essential for murine neural crest proliferation, pharyngeal arch development, and craniofacial morphogenesis
- PMID: 28791750
- PMCID: PMC5690816
- DOI: 10.1002/dvdy.24563
Histone deacetylase 1 and 2 are essential for murine neural crest proliferation, pharyngeal arch development, and craniofacial morphogenesis
Abstract
Background: Craniofacial anomalies involve defective pharyngeal arch development and neural crest function. Copy number variation at 1p35, containing histone deacetylase 1 (Hdac1), or 6q21-22, containing Hdac2, are implicated in patients with craniofacial defects, suggesting an important role in guiding neural crest development. However, the roles of Hdac1 and Hdac2 within neural crest cells remain unknown.
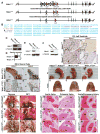
Results: The neural crest and its derivatives express both Hdac1 and Hdac2 during early murine development. Ablation of Hdac1 and Hdac2 within murine neural crest progenitor cells cause severe hemorrhage, atrophic pharyngeal arches, defective head morphogenesis, and complete embryonic lethality. Embryos lacking Hdac1 and Hdac2 in the neural crest exhibit decreased proliferation and increased apoptosis in both the neural tube and the first pharyngeal arch. Mechanistically, loss of Hdac1 and Hdac2 upregulates cyclin-dependent kinase inhibitors Cdkn1a, Cdkn1b, Cdkn1c, Cdkn2b, Cdkn2c, and Tp53 within the first pharyngeal arch.
Conclusions: Our results show that Hdac1 and Hdac2 function redundantly within the neural crest to regulate proliferation and the development of the pharyngeal arches by means of repression of cyclin-dependent kinase inhibitors. Developmental Dynamics 246:1015-1026, 2017. © 2017 Wiley Periodicals, Inc.
Keywords: craniofacial morphogenesis; histone deacetylase; neural crest; pharyngeal arch.
© 2017 Wiley Periodicals, Inc.
Figures



Similar articles
-
A single allele of Hdac2 but not Hdac1 is sufficient for normal mouse brain development in the absence of its paralog.Development. 2014 Feb;141(3):604-616. doi: 10.1242/dev.100487. Development. 2014. PMID: 24449838 Free PMC article.
-
Distinct Roles for Intestinal Epithelial Cell-Specific Hdac1 and Hdac2 in the Regulation of Murine Intestinal Homeostasis.J Cell Physiol. 2016 Feb;231(2):436-48. doi: 10.1002/jcp.25090. J Cell Physiol. 2016. PMID: 26174178
-
HDAC1 and HDAC2 control the specification of neural crest cells into peripheral glia.J Neurosci. 2014 Apr 23;34(17):6112-22. doi: 10.1523/JNEUROSCI.5212-13.2014. J Neurosci. 2014. PMID: 24760871 Free PMC article.
-
HDAC1 and HDAC2 in mouse oocytes and preimplantation embryos: Specificity versus compensation.Cell Death Differ. 2016 Jul;23(7):1119-27. doi: 10.1038/cdd.2016.31. Epub 2016 Apr 15. Cell Death Differ. 2016. PMID: 27082454 Free PMC article. Review.
-
The role of foxi family transcription factors in the development of the ear and jaw.Curr Top Dev Biol. 2015;111:461-95. doi: 10.1016/bs.ctdb.2014.11.014. Epub 2015 Jan 21. Curr Top Dev Biol. 2015. PMID: 25662269 Free PMC article. Review.
Cited by
-
Histone Epigenetic Signatures in Embryonic Limb Interdigital Cells Fated to Die.Cells. 2021 Apr 15;10(4):911. doi: 10.3390/cells10040911. Cells. 2021. PMID: 33921015 Free PMC article.
-
Epigenetics and the evolution of form: Experimental manipulation of a chromatin modification causes species-specific changes to the craniofacial skeleton.Evol Dev. 2024 Jan;26(1):e12461. doi: 10.1111/ede.12461. Epub 2023 Oct 18. Evol Dev. 2024. PMID: 37850843 Free PMC article.
-
Searching for Essential Genes and Targeted Drugs Common to Breast Cancer and Osteoarthritis.Comb Chem High Throughput Screen. 2024;27(2):238-255. doi: 10.2174/1386207326666230508113036. Comb Chem High Throughput Screen. 2024. PMID: 37157194
-
Molecular Regulation of Palatogenesis and Clefting: An Integrative Analysis of Genetic, Epigenetic Networks, and Environmental Interactions.Int J Mol Sci. 2025 Feb 6;26(3):1382. doi: 10.3390/ijms26031382. Int J Mol Sci. 2025. PMID: 39941150 Free PMC article. Review.
-
Pathological MAPK activation-mediated lymphatic basement membrane disruption causes lymphangiectasia that is treatable with ravoxertinib.JCI Insight. 2022 Sep 8;7(17):e153033. doi: 10.1172/jci.insight.153033. JCI Insight. 2022. PMID: 36073544 Free PMC article.
References
-
- Amaral JD, Xavier JM, Steer CJ, Rodrigues CM. The role of p53 in apoptosis. Discov Med. 2010;9:145–152. - PubMed
-
- Benson MT, Dalen K, Mancuso AA, Kerr HH, Cacciarelli AA, Mafee MF. Congenital anomalies of the branchial apparatus: embryology and pathologic anatomy. Radiographics. 1992;12:943–960. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

