Peripheral Elevation of a Klotho Fragment Enhances Brain Function and Resilience in Young, Aging, and α-Synuclein Transgenic Mice
- PMID: 28793260
- PMCID: PMC5816951
- DOI: 10.1016/j.celrep.2017.07.024
Peripheral Elevation of a Klotho Fragment Enhances Brain Function and Resilience in Young, Aging, and α-Synuclein Transgenic Mice
Abstract
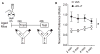
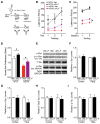
Cognitive dysfunction and decreased mobility from aging and neurodegenerative conditions, such as Parkinson and Alzheimer diseases, are major biomedical challenges in need of more effective therapies. Increasing brain resilience may represent a new treatment strategy. Klotho, a longevity factor, enhances cognition when genetically and broadly overexpressed in its full, wild-type form over the mouse lifespan. Whether acute klotho treatment can rapidly enhance cognitive and motor functions or induce resilience is a gap in our knowledge of its therapeutic potential. Here, we show that an α-klotho protein fragment (αKL-F), administered peripherally, surprisingly induced cognitive enhancement and neural resilience despite impermeability to the blood-brain barrier in young, aging, and transgenic α-synuclein mice. αKL-F treatment induced cleavage of the NMDAR subunit GluN2B and also enhanced NMDAR-dependent synaptic plasticity. GluN2B blockade abolished αKL-F-mediated effects. Peripheral αKL-F treatment is sufficient to induce neural enhancement and resilience in mice and may prove therapeutic in humans.
Keywords: Alzheimer disease; NMDA receptors; Parkinson disease; aging; brain; cognition; klotho; mice; motor function; α-synuclein.
Copyright © 2017 The Author(s). Published by Elsevier Inc. All rights reserved.
Figures


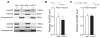


References
-
- Almeida OP, Morar B, Hankey GJ, Yeap BB, Golledge J, Jablensky A, Flicker L. Longevity Klotho gene polymorphism and the risk of dementia in older men. Maturitas. 2017;101:1–5. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

