Viscous drag on the flagellum activates Bacillus subtilis entry into the K-state
- PMID: 28800172
- PMCID: PMC5653444
- DOI: 10.1111/mmi.13770
Viscous drag on the flagellum activates Bacillus subtilis entry into the K-state
Abstract
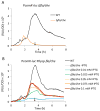
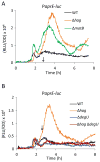
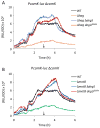
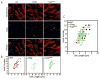
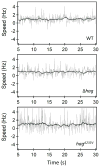
Bacillus subtilis flagella are not only required for locomotion but also act as sensors that monitor environmental changes. Although how the signal transmission takes place is poorly understood, it has been shown that flagella play an important role in surface sensing by transmitting a mechanical signal to control the DegS-DegU two-component system. Here we report a role for flagella in the regulation of the K-state, which enables transformability and antibiotic tolerance (persistence). Mutations impairing flagellar synthesis are inferred to increase DegU-P, which inhibits the expression of ComK, the master regulator for the K-state, and reduces transformability. Tellingly, both deletion of the flagellin gene and straight filament (hagA233V ) mutations increased DegU phosphorylation despite the fact that both mutants had wild type numbers of basal bodies and the flagellar motors were functional. We propose that higher viscous loads on flagellar motors result in lower DegU-P levels through an unknown signaling mechanism. This flagellar-load based mechanism ensures that cells in the motile subpopulation have a tenfold enhanced likelihood of entering the K-state and taking up DNA from the environment. Further, our results suggest that the developmental states of motility and competence are related and most commonly occur in the same epigenetic cell type.
© 2017 John Wiley & Sons Ltd.
Figures


Similar articles
-
Impaired competence in flagellar mutants of Bacillus subtilis is connected to the regulatory network governed by DegU.Environ Microbiol Rep. 2018 Feb;10(1):23-32. doi: 10.1111/1758-2229.12601. Epub 2017 Dec 4. Environ Microbiol Rep. 2018. PMID: 29124898
-
DegU-P represses expression of the motility fla-che operon in Bacillus subtilis.J Bacteriol. 2004 Sep;186(18):6003-14. doi: 10.1128/JB.186.18.6003-6014.2004. J Bacteriol. 2004. PMID: 15342569 Free PMC article.
-
A mechanical signal transmitted by the flagellum controls signalling in Bacillus subtilis.Mol Microbiol. 2013 Oct;90(1):6-21. doi: 10.1111/mmi.12342. Epub 2013 Aug 14. Mol Microbiol. 2013. PMID: 23888912 Free PMC article.
-
The structure and regulation of flagella in Bacillus subtilis.Annu Rev Genet. 2014;48:319-40. doi: 10.1146/annurev-genet-120213-092406. Epub 2014 Sep 10. Annu Rev Genet. 2014. PMID: 25251856 Free PMC article. Review.
-
Swarming motility: it better be wet.Curr Biol. 2005 Aug 9;15(15):R599-600. doi: 10.1016/j.cub.2005.07.042. Curr Biol. 2005. PMID: 16085482 Review.
Cited by
-
The sprT Gene of Bacillus velezensis FZB42 Is Involved in Biofilm Formation and Bacilysin Production.Int J Mol Sci. 2023 Nov 27;24(23):16815. doi: 10.3390/ijms242316815. Int J Mol Sci. 2023. PMID: 38069139 Free PMC article.
-
Flagellum-Mediated Mechanosensing and RflP Control Motility State of Pathogenic Escherichia coli.mBio. 2020 Mar 24;11(2):e02269-19. doi: 10.1128/mBio.02269-19. mBio. 2020. PMID: 32209689 Free PMC article.
-
Citrate Supplementation Modulates Medium Viscosity and Poly-γ-Glutamic Acid Synthesis by Engineered B. subtilis 168.Eng Life Sci. 2025 Mar 4;25(3):e70009. doi: 10.1002/elsc.70009. eCollection 2025 Mar. Eng Life Sci. 2025. PMID: 40046170 Free PMC article.
-
c-di-GMP-Dependent Regulation of Motility by comFB and comFC.bioRxiv [Preprint]. 2025 Jul 11:2025.07.11.664319. doi: 10.1101/2025.07.11.664319. bioRxiv. 2025. PMID: 40672238 Free PMC article. Preprint.
-
Roadmap on emerging concepts in the physical biology of bacterial biofilms: from surface sensing to community formation.Phys Biol. 2021 Jun 23;18(5):10.1088/1478-3975/abdc0e. doi: 10.1088/1478-3975/abdc0e. Phys Biol. 2021. PMID: 33462162 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

