Genotypes of hepatitis a virus in Turkey: first report and clinical profile of children infected with sub-genotypes IA and IIIA
- PMID: 28800730
- PMCID: PMC5553755
- DOI: 10.1186/s12879-017-2667-3
Genotypes of hepatitis a virus in Turkey: first report and clinical profile of children infected with sub-genotypes IA and IIIA
Abstract
Background: Hepatitis A virus (HAV) is a food and water-borne virus causing clinical (mainly hepatitis) and subclinical disease in humans. It is important to characterize circulating strains of HAV in order to prevent HAV infections using efficacious vaccines. The aim of this study was the detection and characterization of the circulating strains of HAV in Turkey by performing serology, RT-PCR, sequencing and phylogenetic analysis.
Methods: In this study, 355 HAV suspected cases were analysed by ELISA for the presence of antibodies to HAV. RNA was extracted from 54 HAV IgM positive human sera. None of the suspect cases were vaccinated against HAV and they never received blood transfusions. Samples found positive by RT-PCR using primers targeting the VP1/VP2A junction and VP1/VP3 capsid region of HAV, were subjected to sequencing and phylogenetic analyses.
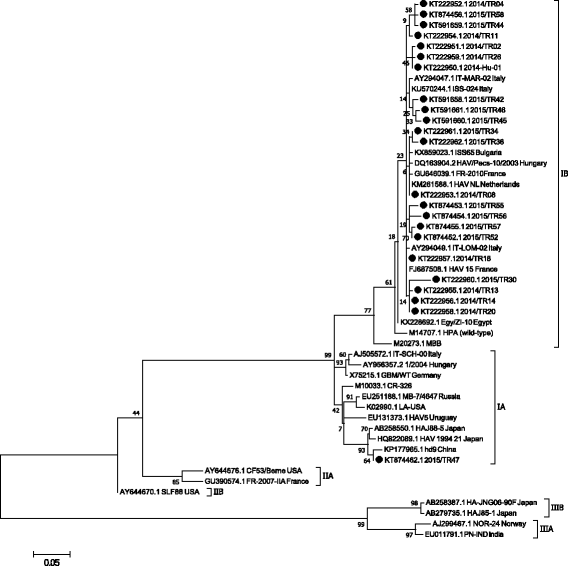
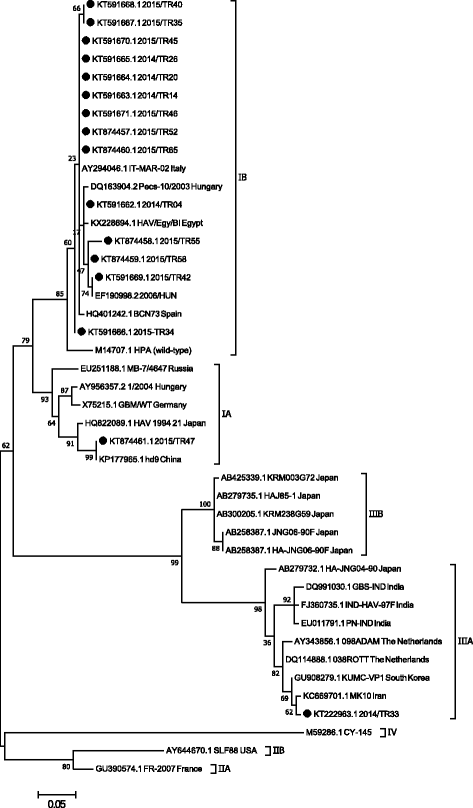
Results: IgM type antibodies to HAV were detected in 54 patients. Twenty one of them were students. The age of IgM positive cases was between 3 and 60 years. IgM positivity differed in age groups and was higher in the age group 3 to 10 years. Phylogenetic analysis showed that the majority of HAV strains detected in this study belong to the "HAV 1B" cluster. In addition, the HAV sub-genotypes IA (KT874461.1) and IIIA (KT222963.1) were found in 2 children. These sub-genotypes were not previously reported in Turkey. The child who carried sub-genotype IIIA travelled to Afghanistan and presented with abdominal pain, icterus and vomitus. He was positive for anti-HAV IgM and IgG but negative for hepatitis B and C. Liver enzymes like aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, gamma-glutamyl transferase and lactate dehydrogenase were severely elevated. Bilirubin levels were also increased. White blood cells, neutrophils and hemoglobin were decreased while lymphocytes and monocytes were increased. Similar clinical signs and laboratory findings were reported for the child infected with sub-genotype IA but aspartate aminotransferase and alanine aminotransferase were not severely elevated.
Conclusions: The results indicate that molecular studies determining the HAV genotype variation in Turkey are timely and warranted. The majority of IgM positive cases in 3-10 year old patients indicate that childhood vaccination is important. Sub-genotype IB is the most prevalant genotype in Turkey. Surprisingly, sub-genotype IA and IIIA are also present in Turkey; future diagnostic efforts need to include diagnostic methods which can identify this emerging HAV genotypes. Our results also show that one important risk factor for contracting hepatitis A virus is international travel since genotype IIIA was detected in a child who had travelled to Afghanistan.
Keywords: Hepatitis a virus sequencing; Phylogenetic; Turkey; VP1/VP2A; VP1/VP3.
Conflict of interest statement
Ethics approval and consent to participate
Ethical approval was taken from the Ethics Committee of the University of Istanbul. Human included in the study were informed and asked to participate in the study. Consent was taken from patients.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
-
- Fiore AE, Wasley A, Bell BP. Advisory Committee on Immunization Practices (ACIP), Prevention of hepatitis A through active or passive immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). The MMWR Recommendations and Reports. 2006;55(RR-7):1–23. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

