Reduction of Mosquito Survival in Mice Vaccinated with Anopheles stephensi Glucose Transporter
- PMID: 28804714
- PMCID: PMC5540378
- DOI: 10.1155/2017/3428186
Reduction of Mosquito Survival in Mice Vaccinated with Anopheles stephensi Glucose Transporter
Abstract
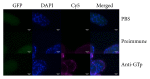
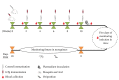
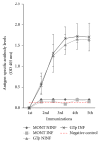
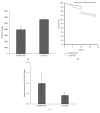
Despite the fact that recent efforts to control/eradicate malaria have contributed to a significant decrease in the number of cases and deaths, the disease remains a global health challenge. Vaccines based on mosquito salivary gland antigens are a potential approach for reducing vector populations and malaria parasites. The Anopheles AGAP007752 gene encodes for a glucose transporter that is upregulated during Plasmodium infection, and its knockdown decreases the number of sporozoites in mosquito salivary glands. These results together with the fact that glucose is a vital source of energy suggested that a glucose transporter is a candidate protective antigen for the control of mosquito infestations and Plasmodium infection. To address this hypothesis, herein we investigate the effect of mice vaccination with an immunogenic peptide from mosquito glucose transporter on Anopheles stephensi fitness and Plasmodium berghei infection. We showed that vaccination with a peptide of glucose transporter reduced mosquito survival by 5% when compared to controls. However, the reduction in Plasmodium infection was not significant in mosquitoes fed on vaccinated mice. The effect of the peptide vaccination on mosquito survival is important to reduce infestation by malaria vectors. These results support further research on developing glucose transporter-based vaccines to reduce mosquito fitness.
Figures
Similar articles
-
Immunomodulation by Mosquito Salivary Protein AgSAP Contributes to Early Host Infection by Plasmodium.mBio. 2021 Dec 21;12(6):e0309121. doi: 10.1128/mBio.03091-21. Epub 2021 Dec 14. mBio. 2021. PMID: 34903042 Free PMC article.
-
Gene expression changes in the salivary glands of Anopheles coluzzii elicited by Plasmodium berghei infection.Parasit Vectors. 2015 Sep 23;8:485. doi: 10.1186/s13071-015-1079-8. Parasit Vectors. 2015. PMID: 26395987 Free PMC article.
-
Solute carriers affect Anopheles stephensi survival and Plasmodium berghei infection in the salivary glands.Sci Rep. 2017 Jul 21;7(1):6141. doi: 10.1038/s41598-017-06317-6. Sci Rep. 2017. PMID: 28733628 Free PMC article.
-
NF-κB-Like Signaling Pathway REL2 in Immune Defenses of the Malaria Vector Anopheles gambiae.Front Cell Infect Microbiol. 2017 Jun 21;7:258. doi: 10.3389/fcimb.2017.00258. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 28680852 Free PMC article. Review.
-
How does Anopheles gambiae kill malaria parasites?Parassitologia. 1999 Sep;41(1-3):169-75. Parassitologia. 1999. PMID: 10697851 Review.
Cited by
-
Quantitative Proteomics Identifies Metabolic Pathways Affected by Babesia Infection and Blood Feeding in the Sialoproteome of the Vector Rhipicephalus bursa.Vaccines (Basel). 2020 Feb 19;8(1):91. doi: 10.3390/vaccines8010091. Vaccines (Basel). 2020. PMID: 32092882 Free PMC article.
-
Glucose transporter GLUT1 influences Plasmodium berghei infection in Anopheles stephensi.Parasit Vectors. 2020 Jun 5;13(1):285. doi: 10.1186/s13071-020-04155-6. Parasit Vectors. 2020. PMID: 32503601 Free PMC article.
-
Mining gene expression data for rational identification of novel drug targets and vaccine candidates against the cattle tick, Rhipicephalus microplus.Exp Appl Acarol. 2023 Oct;91(2):291-317. doi: 10.1007/s10493-023-00838-8. Epub 2023 Sep 27. Exp Appl Acarol. 2023. PMID: 37755526 Free PMC article.
References
-
- WHO. World Malaria Report 2016. 2016, http://apps.who.int/iris/bitstream/10665/252038/1/9789241511711-eng.pdf?....
-
- WHO. World Malaria Report 2015. 2015, http://apps.who.int/iris/bitstream/10665/200018/1/9789241565158_eng.pdf?....
-
- Agnandji S. T., Lell B., Fernandes J. F., et al. Efficacy and safety of the RTS,S/AS01 malaria vaccine during 18 months after vaccination: a phase 3 randomized, controlled trial in children and young infants at 11 African sites. PLoS Medicine. 2014;11(7) doi: 10.1371/journal.pmed.1001685.e1001685 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

