Integrated analysis of mRNA-seq and miRNA-seq reveals the potential roles of sex-biased miRNA-mRNA pairs in gonad tissue of dark sleeper (Odontobutis potamophila)
- PMID: 28806919
- PMCID: PMC5557427
- DOI: 10.1186/s12864-017-3995-9
Integrated analysis of mRNA-seq and miRNA-seq reveals the potential roles of sex-biased miRNA-mRNA pairs in gonad tissue of dark sleeper (Odontobutis potamophila)
Abstract
Background: The dark sleeper (Odontobutis potamophila) is an important commercial fish species which shows a sexually dimorphic growth pattern. However, the lack of sex transcriptomic data is hindering further research and genetically selective breeding of the dark sleeper. In this study, integrated analysis of mRNA and miRNA was performed on gonad tissue to elucidate the molecular mechanisms of sex determination and differentiation in the dark sleeper.
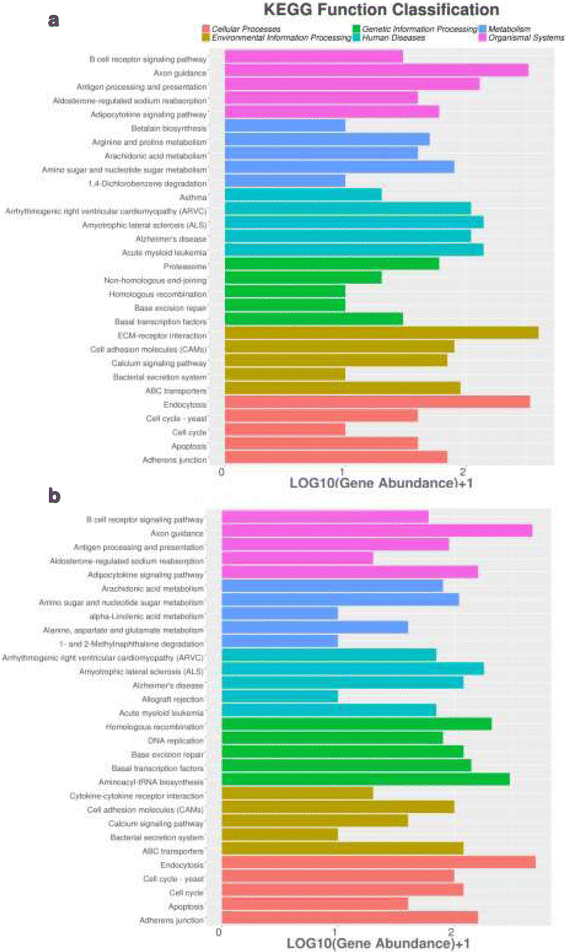
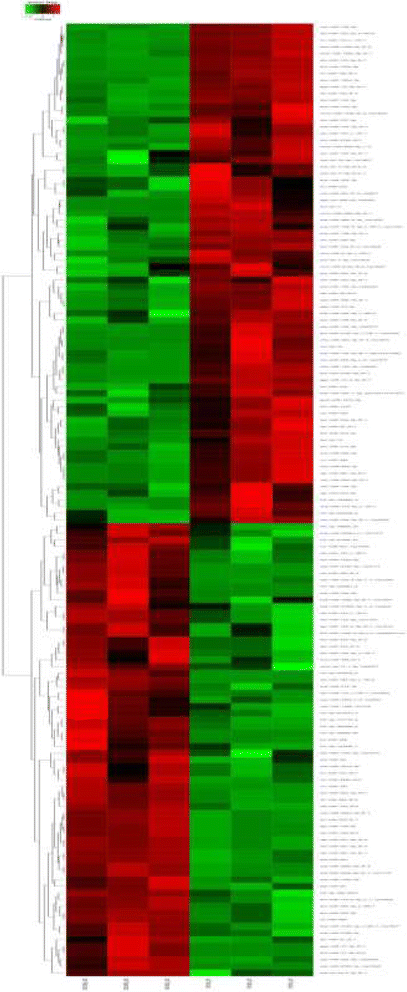
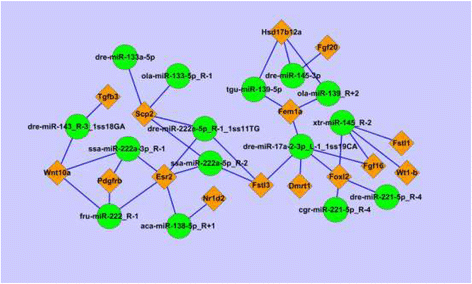
Results: A total of 143 differentially expressed miRNAs and 16,540 differentially expressed genes were identified. Of these, 8103 mRNAs and 75 miRNAs were upregulated in testes, and 8437 mRNAs and 68 miRNAs were upregulated in ovaries. Integrated analysis of miRNA and mRNA expression profiles predicted more than 50,000 miRNA-mRNA interaction sites, and among them 27,583 negative miRNA-mRNA interactions. A number of sex related genes were targeted by sex-biased miRNAs. The relationship between 15 sex-biased genes and 15 sex-biased miRNAs verified by using qRT-PCR were described. Additionally, a number of SNPs were revealed through the transcriptome data.
Conclusions: The overall results of this study facilitate our understanding of the molecular mechanism underlying sex determination and differentiation and provide valuable genomic information for selective breeding of the dark sleeper.
Keywords: Dark sleeper; Gonads; Sex determination; Sex differentiation; mRNA; miRNA.
Conflict of interest statement
Ethics approval and consent to participate
All experiments were performed according to the Guidelines for the Care and Use of Laboratory Animals in China. This study was also approved by the Ethics Committee for Experimental Animals at Nanjing Normal University (Permit 91, No. SYXK2015–0028).
Consent for publication
Not applicable.
Competing interests
The authors declare no conflict interests of this article.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures


Similar articles
-
Integrated mRNA and miRNA expression profile analyses reveal the potential roles of sex-biased miRNA-mRNA pairs in gonad tissues of the Chinese concave-eared torrent frog (Odorrana tormota).J Exp Zool B Mol Dev Evol. 2019 May;332(3-4):69-80. doi: 10.1002/jez.b.22851. Epub 2019 Apr 9. J Exp Zool B Mol Dev Evol. 2019. PMID: 30964604
-
Identification and differential expression of microRNAs in testis and ovary of Amur sturgeon (Acipenser schrenckii).Gene. 2018 Jun 5;658:36-46. doi: 10.1016/j.gene.2018.03.014. Epub 2018 Mar 7. Gene. 2018. PMID: 29524574
-
Evolutionary conservation and divergence of Vasa, Dazl and Nanos1 during embryogenesis and gametogenesis in dark sleeper (Odontobutis potamophila).Gene. 2018 Sep 25;672:21-33. doi: 10.1016/j.gene.2018.06.016. Epub 2018 Jun 6. Gene. 2018. PMID: 29885464
-
Integrated analysis of miRNA and mRNA expression profiles in tilapia gonads at an early stage of sex differentiation.BMC Genomics. 2016 May 4;17:328. doi: 10.1186/s12864-016-2636-z. BMC Genomics. 2016. PMID: 27142172 Free PMC article.
-
Single cell transcriptome sequencing: A new approach for the study of mammalian sex determination.Mol Cell Endocrinol. 2018 Jun 15;468:11-18. doi: 10.1016/j.mce.2018.01.013. Epub 2018 Jan 31. Mol Cell Endocrinol. 2018. PMID: 29371022 Review.
Cited by
-
Integrated Analysis of mRNA and miRNA Associated with Reproduction in Female and Male Gonads in Abalone (Haliotis discus hannai).Int J Mol Sci. 2025 Mar 31;26(7):3235. doi: 10.3390/ijms26073235. Int J Mol Sci. 2025. PMID: 40244070 Free PMC article.
-
Transcriptional reprogramming strategies and miRNA-mediated regulation networks of Taxus media induced into callus cells from tissues.BMC Genomics. 2020 Feb 18;21(1):168. doi: 10.1186/s12864-020-6576-2. BMC Genomics. 2020. PMID: 32070278 Free PMC article.
-
Gonadal miRNomes and transcriptomes in infected fish reveal sexually dimorphic patterns of the immune response.Funct Integr Genomics. 2025 Jan 30;25(1):29. doi: 10.1007/s10142-025-01537-w. Funct Integr Genomics. 2025. PMID: 39883212 Free PMC article.
-
Integrated Analysis of mRNA- and miRNA-Seq in the Ovary of Rare Minnow Gobiocypris rarus in Response to 17α-Methyltestosterone.Front Genet. 2021 Aug 5;12:695699. doi: 10.3389/fgene.2021.695699. eCollection 2021. Front Genet. 2021. PMID: 34421998 Free PMC article.
-
Effect of quercetin on muscle growth and antioxidant status of the dark sleeper Odontobutis potamophila.Front Genet. 2022 Jul 25;13:938526. doi: 10.3389/fgene.2022.938526. eCollection 2022. Front Genet. 2022. PMID: 35957695 Free PMC article.
References
-
- Adolfi MC, Carreira AC, Jesus LW, Bogerd J, Funes RM, Schartl M, Sogayar MC, Borella MI. Molecular cloning and expression analysis of dmrt1 and sox9 during gonad development and male reproductive cycle in the lambari fish. Astyanax altiparanae Reprod Biol Endocrinol. 2015;13:2. doi: 10.1186/1477-7827-13-2. - DOI - PMC - PubMed
-
- Kamiya T, Kai W, Tasumi S, Oka A, Matsunaga T, Mizuno N, Fujita M, Suetake H, Suzuki S, Hosoya S, et al. A trans-species missense SNP in Amhr2 is associated with sex determination in the tiger pufferfish, Takifugu rubripes (fugu) PLoS Genet. 2012;8(7) doi: 10.1371/journal.pgen.1002798. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

