Regulation of L-type CaV1.3 channel activity and insulin secretion by the cGMP-PKG signaling pathway
- PMID: 28807144
- PMCID: PMC5776030
- DOI: 10.1016/j.ceca.2017.05.008
Regulation of L-type CaV1.3 channel activity and insulin secretion by the cGMP-PKG signaling pathway
Abstract
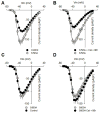
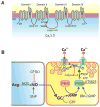
cGMP is a second messenger widely used in the nervous system and other tissues. One of the major effectors for cGMP is the serine/threonine protein kinase, cGMP-dependent protein kinase (PKG), which catalyzes the phosphorylation of a variety of proteins including ion channels. Previously, it has been shown that the cGMP-PKG signaling pathway inhibits Ca2+ currents in rat vestibular hair cells and chromaffin cells. This current allegedly flow through voltage-gated CaV1.3L-type Ca2+ channels, and is important for controlling vestibular hair cell sensory function and catecholamine secretion, respectively. Here, we show that native L-type channels in the insulin-secreting RIN-m5F cell line, and recombinant CaV1.3 channels heterologously expressed in HEK-293 cells, are regulatory targets of the cGMP-PKG signaling cascade. Our results indicate that the CaVα1 ion-conducting subunit of the CaV1.3 channels is highly expressed in RIN-m5F cells and that the application of 8-Br-cGMP, a membrane-permeable analogue of cGMP, significantly inhibits Ca2+ macroscopic currents and impair insulin release stimulated with high K+. In addition, KT-5823, a specific inhibitor of PKG, prevents the current inhibition generated by 8-Br-cGMP in the heterologous expression system. Interestingly, mutating the putative phosphorylation sites to residues resistant to phosphorylation showed that the relevant PKG sites for CaV1.3 L-type channel regulation centers on two amino acid residues, Ser793 and Ser860, located in the intracellular loop connecting the II and III repeats of the CaVα1 pore-forming subunit of the channel. These findings unveil a novel mechanism for how the cGMP-PKG signaling pathway may regulate CaV1.3 channels and contribute to regulate insulin secretion.
Keywords: Cav channels; Insulin; L-type channels; PKG; Rin-m5F cells; cGMP.
Copyright © 2017 Elsevier Ltd. All rights reserved.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
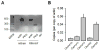
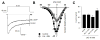
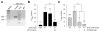
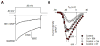
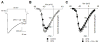
Similar articles
-
Modulation of Kv3 potassium channels expressed in CHO cells by a nitric oxide-activated phosphatase.J Physiol. 2001 Feb 1;530(Pt 3):345-58. doi: 10.1111/j.1469-7793.2001.0345k.x. J Physiol. 2001. PMID: 11281123 Free PMC article.
-
cGMP-dependent protein kinase mediates stimulation of L-type calcium current by cGMP in rabbit atrial cells.Cardiovasc Res. 2000 Nov;48(2):310-22. doi: 10.1016/s0008-6363(00)00178-4. Cardiovasc Res. 2000. PMID: 11054477
-
Protein kinase G phosphorylates Cav1.2 alpha1c and beta2 subunits.Circ Res. 2007 Aug 31;101(5):465-74. doi: 10.1161/CIRCRESAHA.107.156976. Epub 2007 Jul 12. Circ Res. 2007. PMID: 17626895
-
Species- and tissue-dependent effects of NO and cyclic GMP on cardiac ion channels.Comp Biochem Physiol A Mol Integr Physiol. 2005 Oct;142(2):136-43. doi: 10.1016/j.cbpb.2005.04.012. Epub 2005 May 31. Comp Biochem Physiol A Mol Integr Physiol. 2005. PMID: 15927494 Review.
-
Cav1.3 and Cav1.2 channels of adrenal chromaffin cells: emerging views on cAMP/cGMP-mediated phosphorylation and role in pacemaking.Biochim Biophys Acta. 2013 Jul;1828(7):1608-18. doi: 10.1016/j.bbamem.2012.11.013. Epub 2012 Nov 15. Biochim Biophys Acta. 2013. PMID: 23159773 Review.
Cited by
-
Epigenetic alternations of microRNAs and DNA methylation contribute to gestational diabetes mellitus.J Cell Mol Med. 2020 Dec;24(23):13899-13912. doi: 10.1111/jcmm.15984. Epub 2020 Oct 21. J Cell Mol Med. 2020. PMID: 33085184 Free PMC article.
-
The Leucine-Rich Repeat Kinase 2 Variant LRRK2G2019S Up-Regulates L-Type (CaV1.3) Calcium Channel via the CaVβ3 Subunit: Possible Role in the Pathogenesis of Parkinson's Disease.Int J Mol Sci. 2025 Mar 31;26(7):3229. doi: 10.3390/ijms26073229. Int J Mol Sci. 2025. PMID: 40244042 Free PMC article.
-
Unveiling the multifaceted role of the FLNC gene: implications for cancer diagnosis and prognosis.Eur J Med Res. 2025 Jul 9;30(1):608. doi: 10.1186/s40001-025-02876-x. Eur J Med Res. 2025. PMID: 40635056 Free PMC article.
-
Potential Mechanism of Dingji Fumai Decoction Against Atrial Fibrillation Based on Network Pharmacology, Molecular Docking, and Experimental Verification Integration Strategy.Front Cardiovasc Med. 2021 Nov 11;8:712398. doi: 10.3389/fcvm.2021.712398. eCollection 2021. Front Cardiovasc Med. 2021. PMID: 34859062 Free PMC article.
-
Activation of mGluR1 negatively modulates glutamate-induced phase shifts of the circadian pacemaker in the mouse suprachiasmatic nucleus.Neurobiol Sleep Circadian Rhythms. 2023 Feb 19;14:100089. doi: 10.1016/j.nbscr.2023.100089. eCollection 2023 May. Neurobiol Sleep Circadian Rhythms. 2023. PMID: 36874931 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

