Bimetallic iron-iron and iron-zinc complexes of the redox-active ONO pincer ligand
- PMID: 28808535
- PMCID: PMC5530940
- DOI: 10.1039/c5sc03006d
Bimetallic iron-iron and iron-zinc complexes of the redox-active ONO pincer ligand
Abstract
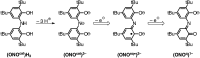
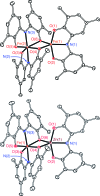
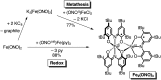
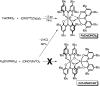
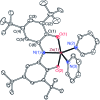
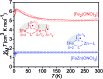
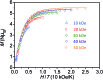
A new bimetallic platform comprising a six-coordinate Fe(ONO)2 unit bound to an (ONO)M (M = Fe, Zn) has been discovered ((ONOcat)H3 = bis(3,5-di-tert-butyl-2-phenol)amine). Reaction of Fe(ONO)2 with either (ONOcat)Fe(py)3 or with (ONOq)FeCl2 under reducing conditions led to the formation of the bimetallic complex Fe2(ONO)3, which includes unique five- and six-coordinate iron centers. Similarly, the reaction of Fe(ONO)2 with the new synthon (ONOsq˙)Zn(py)2 led to the formation of the heterobimetallic complex FeZn(ONO)3, with a six-coordinate iron center and a five-coordinate zinc center. Both bimetallic complexes were characterized by single-crystal X-ray diffraction studies, solid-state magnetic measurements, and multiple spectroscopic techniques. The magnetic data for FeZn(ONO)3 are consistent with a ground state S = 3/2 spin system, generated from a high-spin iron(ii) center that is antiferromagnetically coupled to a single (ONOsq˙)2- radical ligand. In the case of Fe2(ONO)3, the magnetic data revealed a ground state S = 7/2 spin system arising from the interactions of one high-spin iron(ii) center, one high-spin iron(iii) center, and two (ONOsq˙)2- radical ligands.
Figures
References
-
- Tereniak S. J., Carlson R. K., Clouston L. J., Young V. G., Bill E., Maurice R., Chen Y. S., Kim H. J., Gagliardi L., Lu C. C. J. Am. Chem. Soc. 2013;136:1842–1855. - PubMed
- Clouston L. J., Siedschlag R. B., Rudd P. A., Planas N., Hu S., Miller A. D., Gagliardi L., Lu C. C. J. Am. Chem. Soc. 2013;135:13142–13148. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

