The detection of influenza virus at the community pharmacy to improve the management of local residents with influenza or influenza-like disease
- PMID: 28808580
- PMCID: PMC5549291
- DOI: 10.1186/s40780-017-0091-x
The detection of influenza virus at the community pharmacy to improve the management of local residents with influenza or influenza-like disease
Abstract
Background: As of 2014, community pharmacies in Japan are approved by the Ministry of Health, Labour and Welfare to measure lipid panel, HbA1c, glucose, ALT, AST and γ-GTP, but not to screen for influenza virus. We provided influenza virus screening tests at a community pharmacy to triage people with symptoms suggestive of influenza. Participants were given appropriate advice on how to prevent the spread of and safeguard against influenza. We subsequently evaluated the effects of community pharmacy-based influenza virus screening and prevention measures.
Methods: Local residents with symptoms suggestive of influenza participated in this study. Influenza virus screening tests using nasal samples were provided to the pharmacy, and we assessed samples for the presence of influenza virus. The study consisted of a preliminary interview, informed consent, and screening test on Day 1, and mail-in survey on Day 14.
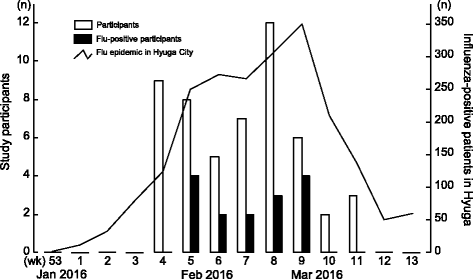
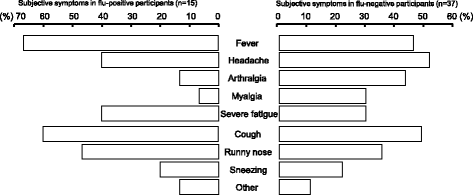
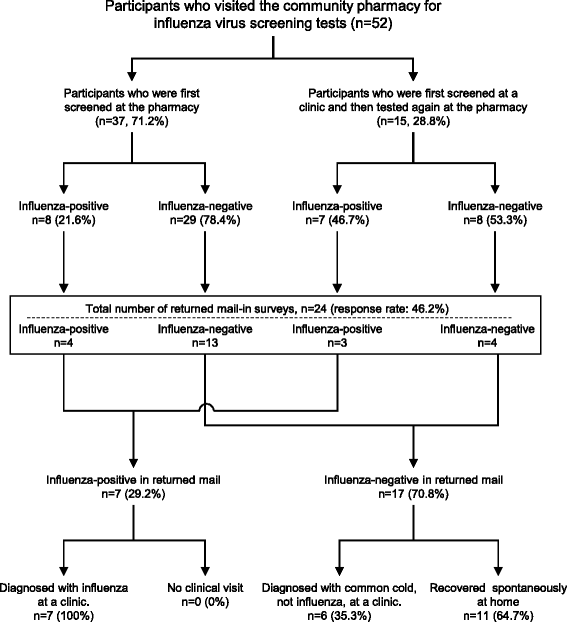
Results: A total 52 local residents participated in the study. The number of participants and influenza virus positive results followed the same trend as the influenza epidemic in the study area. Influenza virus was found in 28.8% of samples. There was no significant difference between the appearance ratios of subjective symptoms among influenza-positive and influenza-negative groups. The percentages of participants who were first screened at the pharmacy, and those who were first screened at a clinic and then tested again at the pharmacy, were 71.2% (37/52) and 28.8% (15/52), respectively. In the latter group, 14 of 15 were negative by screening at the clinic, and one was diagnosed with influenza without testing. Subsequently, 46.8% (7/15) of participants tested positive for influenza by pharmacy-based screening. According to the mail-in survey, all influenza-positive (100%, 7/7) and 35.3% (6/17) of influenza-negative participants visited the clinic after being tested at the community pharmacy; test results between the community pharmacy and clinic were consistent. A total 64.7% (11/17) of symptomatic participants who tested negative recovered spontaneously at home.
Conclusions: Implementation of influenza virus screening followed by provision of appropriate advice for both influenza-positive and influenza-negative participants at the community pharmacy showed a significant effect on improving the health of the local community.
Keywords: Community pharmacy; Influenza; Pharmacist; Point-of-care testing.
Conflict of interest statement
Ethics approval and consent to participate
This study was approved by the ethics committee of Kyushu University of Health and Welfare (No.15–026).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures


Similar articles
-
Evaluation of a community pharmacy-based influenza and group A streptococcal pharyngitis disease management program using polymerase chain reaction point-of-care testing.J Am Pharm Assoc (2003). 2019 Nov-Dec;59(6):872-879. doi: 10.1016/j.japh.2019.07.011. Epub 2019 Aug 29. J Am Pharm Assoc (2003). 2019. PMID: 31474527
-
Community pharmacy rapid influenza A and B screening: A novel approach to expedite patient access to care and improve clinical outcomes.Can Pharm J (Ott). 2016 Mar;149(2):83-9. doi: 10.1177/1715163516629157. Epub 2016 Mar 3. Can Pharm J (Ott). 2016. PMID: 27076819 Free PMC article.
-
Pharmacist-initiated hepatitis C virus screening in a community pharmacy to increase awareness and link to care at the medical center.J Am Pharm Assoc (2003). 2017 May-Jun;57(3S):S259-S264. doi: 10.1016/j.japh.2017.03.006. J Am Pharm Assoc (2003). 2017. PMID: 28506379
-
[Consideration of Advanced Pharmaceutical Control Functions through Pharmacy-provided Home Pharmaceutical Care].Yakugaku Zasshi. 2020;140(7):841-850. doi: 10.1248/yakushi.19-00237-1. Yakugaku Zasshi. 2020. PMID: 32612045 Review. Japanese.
-
[Self-medication Activities in a Community Pharmacy for Student Pharmacist Training].Yakugaku Zasshi. 2016;136(7):951-4. doi: 10.1248/yakushi.15-00257-4. Yakugaku Zasshi. 2016. PMID: 27374956 Review. Japanese.
Cited by
-
Pharmacy-Based Infectious Disease Management Programs Incorporating CLIA-Waived Point-of-Care Tests.J Clin Microbiol. 2020 Apr 23;58(5):e00726-19. doi: 10.1128/JCM.00726-19. Print 2020 Apr 23. J Clin Microbiol. 2020. PMID: 32075903 Free PMC article. Review.
-
Point-of-care testing in private pharmacy and drug retail settings: a narrative review.BMC Infect Dis. 2023 Aug 23;23(1):551. doi: 10.1186/s12879-023-08480-w. BMC Infect Dis. 2023. PMID: 37612636 Free PMC article. Review.
References
-
- Ministry of Health, Labour and Welfare. Notification (No. 0409–4) issued by the Director of Office of Health Policy Bureau. 2014. http://www.mhlw.go.jp/file/06-Seisakujouhou-10800000-Iseikyoku/000009857.... Accessed 3 Apr 2017.
-
- Ministry of Health, Labour and Welfare / National Institute of Infectious Diseases. Infectious Diseases Weekly Report JAPAN, 49th week (Dec 5–11), 18, 2016. http://www0.nih.go.jp/niid/idsc/idwr/IDWR2016/idwr2016-49.pdf. Accessed 3 Apr 2017.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

