Absence of CX3CR1 impairs the internalization of Tau by microglia
- PMID: 28810892
- PMCID: PMC5558740
- DOI: 10.1186/s13024-017-0200-1
Absence of CX3CR1 impairs the internalization of Tau by microglia
Abstract
Background: Extracellular Tau is toxic for neighboring cells, and it contributes to the progression of AD. The CX3CL1/CX3CR1 axis is an important neuron/microglia communication mechanism.
Methods: We studied Tau clearance by microglia both in vitro (microglia primary cultures treated with Cy5-Tau, affinity chromatography to study the binding of Tau to CX3CR1, and Tau-CX3CL1 competition assays) and in vivo (stereotaxic injection of Cy5-Tau into WT and CX3CR1-/- mice). The expression of CX3CR1, CX3CL1 and the microglial phagocytic phenotype were studied in brain tissue samples from AD patients.
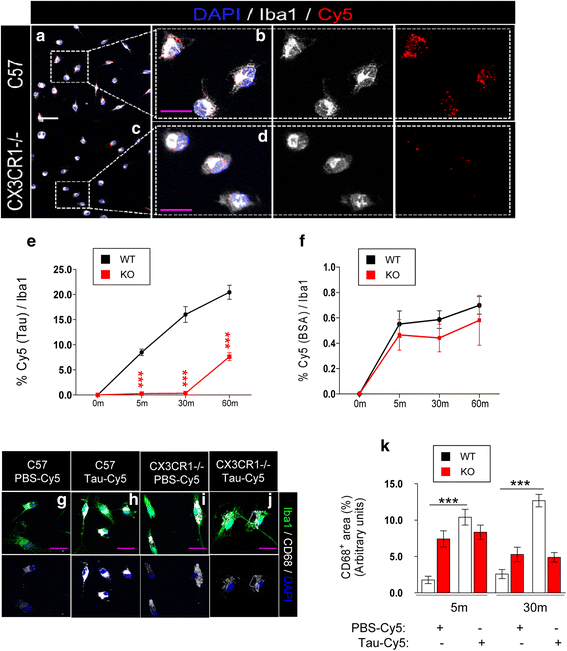
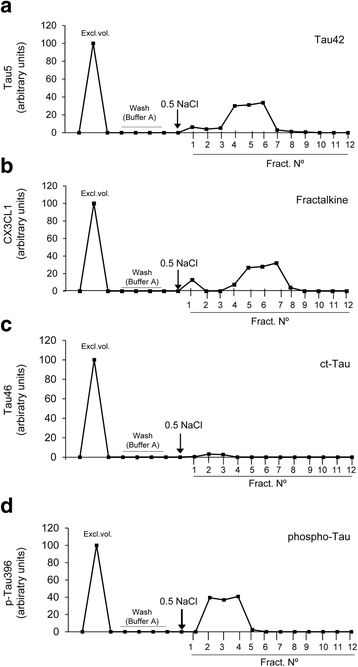
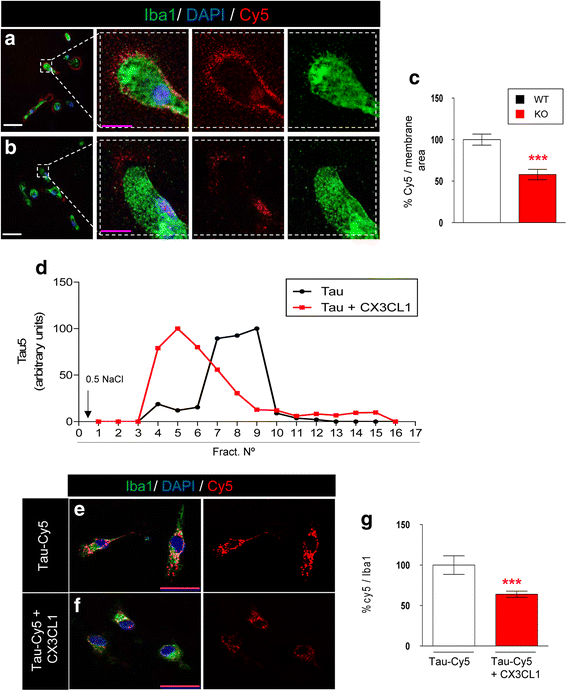
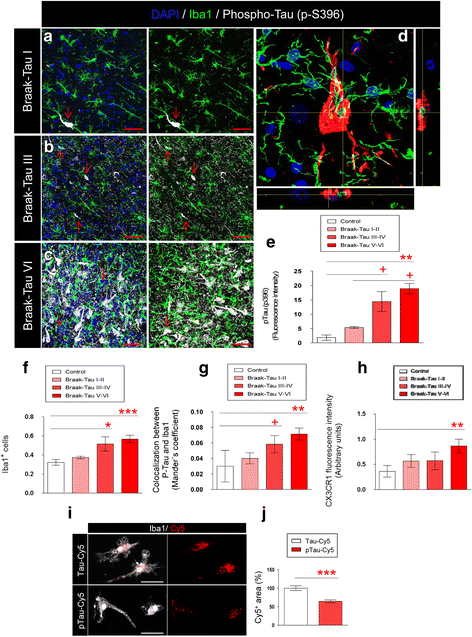
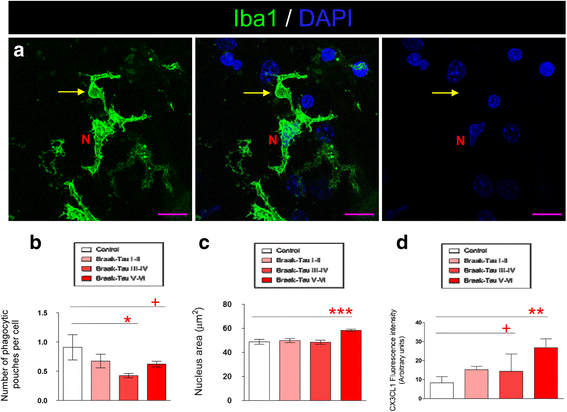
Results: Tau binding to CX3CR1 triggers the internalization of the former by microglia, whereas S396 Tau phosphorylation decreases the binding affinity of this protein to CX3CR1. Of note, the progressive increase in the levels of phosho-Tau occurred in parallel with an increase in CX3CR1. In addition, our studies suggest that the phagocytic capacity of microglia in brain tissue samples from AD patients is decreased. Furthermore, the CX3CR1/CX3CL1 axis may be impaired in late stages of the disease.
Conclusions: Our data suggest that the CX3CR1/CX3CL1 axis plays a key role in the phagocytosis of Tau by microglia in vitro and in vivo and that it is affected as AD progresses. Taken together, our results reveal CX3CR1 as a novel target for the clearance of extracellular Tau.
Keywords: Alzheimer’s disease; CX3CR1; Microglia; Phagocytosis; Tau; Tauopathies.
Conflict of interest statement
Ethics approval
All research involving animals has been approved by the Ethics Board at the CBMSO’s Ethics Committee and the National Ethics Committee (AEEC-CBMSO-62/14). The use of human sample is coordinated by the Banco de Cerebros de la Fundación CIEN; approval reference: CEI PI 29_2016.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

