Direct detection of early-stage cancers using circulating tumor DNA
- PMID: 28814544
- PMCID: PMC6714979
- DOI: 10.1126/scitranslmed.aan2415
Direct detection of early-stage cancers using circulating tumor DNA
Abstract
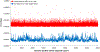
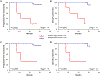
Early detection and intervention are likely to be the most effective means for reducing morbidity and mortality of human cancer. However, development of methods for noninvasive detection of early-stage tumors has remained a challenge. We have developed an approach called targeted error correction sequencing (TEC-Seq) that allows ultrasensitive direct evaluation of sequence changes in circulating cell-free DNA using massively parallel sequencing. We have used this approach to examine 58 cancer-related genes encompassing 81 kb. Analysis of plasma from 44 healthy individuals identified genomic changes related to clonal hematopoiesis in 16% of asymptomatic individuals but no alterations in driver genes related to solid cancers. Evaluation of 200 patients with colorectal, breast, lung, or ovarian cancer detected somatic mutations in the plasma of 71, 59, 59, and 68%, respectively, of patients with stage I or II disease. Analyses of mutations in the circulation revealed high concordance with alterations in the tumors of these patients. In patients with resectable colorectal cancers, higher amounts of preoperative circulating tumor DNA were associated with disease recurrence and decreased overall survival. These analyses provide a broadly applicable approach for noninvasive detection of early-stage tumors that may be useful for screening and management of patients with cancer.
Copyright © 2017 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works.
Figures



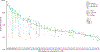
References
-
- Torre LA et al., Global cancer statistics, 2012. CA Cancer J Clin 65, 87 (2015). - PubMed
-
- W. H. Organization, Guide to Cancer Early Diagnosis. Guide to Cancer Early Diagnosis, (2017).
-
- Mazzucchelli R, Colanzi P, Pomante R, Muzzonigro G, Montironi R, Prostate tissue and serum markers. Advances in clinical pathology : the official journal of Adriatic Society of Pathology 4, 111 (2000). - PubMed
-
- Ruibal Morell A, CEA serum levels in non-neoplastic disease. The International journal of biological markers 7, 160 (1992). - PubMed
-
- Galli C, Basso D, Plebani M, CA 19–9: handle with care. Clinical chemistry and laboratory medicine 51, 1369 (2013). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

