Pre-analytic evaluation of volumetric absorptive microsampling and integration in a mass spectrometry-based metabolomics workflow
- PMID: 28815270
- PMCID: PMC5694668
- DOI: 10.1007/s00216-017-0571-8
Pre-analytic evaluation of volumetric absorptive microsampling and integration in a mass spectrometry-based metabolomics workflow
Abstract
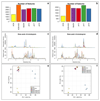
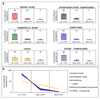
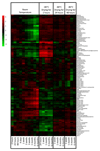
Volumetric absorptive microsampling (VAMS) is a novel approach that allows single-drop (10 μL) blood collection. Integration of VAMS with mass spectrometry (MS)-based untargeted metabolomics is an attractive solution for both human and animal studies. However, to boost the use of VAMS in metabolomics, key pre-analytical questions need to be addressed. Therefore, in this work, we integrated VAMS in a MS-based untargeted metabolomics workflow and investigated pre-analytical strategies such as sample extraction procedures and metabolome stability at different storage conditions. We first evaluated the best extraction procedure for the polar metabolome and found that the highest number and amount of metabolites were recovered upon extraction with acetonitrile/water (70:30). In contrast, basic conditions (pH 9) resulted in divergent metabolite profiles mainly resulting from the extraction of intracellular metabolites originating from red blood cells. In addition, the prolonged storage of blood samples at room temperature caused significant changes in metabolome composition, but once the VAMS devices were stored at - 80 °C, the metabolome remained stable for up to 6 months. The time used for drying the sample did also affect the metabolome. In fact, some metabolites were rapidly degraded or accumulated in the sample during the first 48 h at room temperature, indicating that a longer drying step will significantly change the concentration in the sample. Graphical abstract Volumetric absorptive microsampling (VAMS) is a novel technology that allows single-drop blood collection and, in combination with mass spectrometry (MS)-based untargeted metabolomics, represents an attractive solution for both human and animal studies. In this work, we integrated VAMS in a MS-based untargeted metabolomics workflow and investigated pre-analytical strategies such as sample extraction procedures and metabolome stability at different storage conditions. The latter revealed that prolonged storage of blood samples at room temperature caused significant changes in metabolome composition, but if VAMS devices were stored at - 80 °C, the metabolome remained stable for up to 6 months.
Keywords: Mass spectrometry; Metabolomics; Volumetric absorptive microsampling.
Conflict of interest statement
Figures



References
-
- Guthrie R, Susi A. A simple phenylalanine method for detecting phenylketonuria in large populations of newborn infants. Pediatrics. 1963;32:338–43. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

