Differential Desensitization Observed at Multiple Effectors of Somatic μ-Opioid Receptors Underlies Sustained Agonist-Mediated Inhibition of Proopiomelanocortin Neuron Activity
- PMID: 28821664
- PMCID: PMC5588460
- DOI: 10.1523/JNEUROSCI.1030-17.2017
Differential Desensitization Observed at Multiple Effectors of Somatic μ-Opioid Receptors Underlies Sustained Agonist-Mediated Inhibition of Proopiomelanocortin Neuron Activity
Abstract
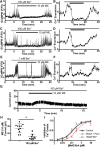
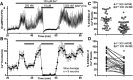
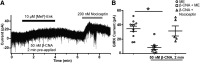
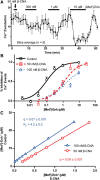
Activation of somatic μ-opioid receptors (MORs) in hypothalamic proopiomelanocortin (POMC) neurons leads to the activation of G-protein-coupled inward rectifier potassium (GIRK) channels and hyperpolarization, but in response to continued signaling MORs undergo acute desensitization resulting in robust reduction in the peak GIRK current after minutes of agonist exposure. We hypothesized that the attenuation of the GIRK current would lead to a recovery of neuronal excitability whereby desensitization of the receptor would lead to a new steady state of POMC neuron activity reflecting the sustained GIRK current observed after the initial decline from peak with continued agonist exposure. However, electrophysiologic recordings and GCaMP6f Ca2+ imaging in POMC neurons in mouse brain slices indicate that maximal inhibition of cellular activity by these measures can be maintained after the GIRK current declines. Blockade of the GIRK current by Ba2+ or Tertiapin-Q did not disrupt the sustained inhibition of Ca2+ transients in the continued presence of agonist, indicating the activation of an effector other than GIRK channels. Use of an irreversible MOR antagonist and Furchgott analysis revealed a low receptor reserve for the activation of GIRK channels but a >90% receptor reserve for the inhibition of Ca2+ events. Altogether, the data show that somatodendritic MORs in POMC neurons inhibit neuronal activity through at least two effectors with distinct levels of receptor reserve and that differentially reflect receptor desensitization. Thus, in POMC cells, the decline in the GIRK current during prolonged MOR agonist exposure does not reflect an increase in cellular activity as expected.SIGNIFICANCE STATEMENT Desensitization of the μ-opioid receptor (MOR) is thought to underlie the development of cellular tolerance to opiate therapy. The present studies focused on MOR desensitization in hypothalamic proopiomelanocortin (POMC) neurons as these neurons produce the endogenous opioid β-endorphin and are heavily regulated by opioids. Prolonged activation of somatic MORs in POMC neurons robustly inhibited action potential firing and Ca2+ activity despite desensitization of the MOR and reduced activation of a potassium current over the same time course. The data show that somatic MORs in POMC neurons couple to multiple effectors that have differential sensitivity to desensitization of the receptor. Thus, in these cells, the cellular consequence of MOR desensitization cannot be defined by the activity of a single effector system.
Keywords: GIRK; action potential; calcium imaging; electrophysiology; hypothalamus; receptor reserve.
Copyright © 2017 the authors 0270-6474/17/378667-11$15.00/0.
Figures


Similar articles
-
Mu Receptors.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31855381 Free Books & Documents.
-
Mu-opioid receptor and delta-opioid receptor differentially regulate microglial inflammatory response to control proopiomelanocortin neuronal apoptosis in the hypothalamus: effects of neonatal alcohol.J Neuroinflammation. 2017 Apr 14;14(1):83. doi: 10.1186/s12974-017-0844-3. J Neuroinflammation. 2017. PMID: 28407740 Free PMC article.
-
Two distinct forms of desensitization of G-protein coupled inwardly rectifying potassium currents evoked by alkaloid and peptide mu-opioid receptor agonists.Mol Cell Neurosci. 2003 Oct;24(2):517-23. doi: 10.1016/s1044-7431(03)00173-8. Mol Cell Neurosci. 2003. PMID: 14572471
-
Rapid effects of estrogen on G protein-coupled receptor activation of potassium channels in the central nervous system (CNS).J Steroid Biochem Mol Biol. 2002 Dec;83(1-5):187-93. doi: 10.1016/s0960-0760(02)00249-2. J Steroid Biochem Mol Biol. 2002. PMID: 12650715 Review.
-
Estrogen modulation of K(+) channel activity in hypothalamic neurons involved in the control of the reproductive axis.Steroids. 2002 May;67(6):447-56. doi: 10.1016/s0039-128x(01)00181-7. Steroids. 2002. PMID: 11960620 Review.
Cited by
-
A Receptor Model With Binding Affinity, Activation Efficacy, and Signal Amplification Parameters for Complex Fractional Response Versus Occupancy Data.Front Pharmacol. 2019 Jun 11;10:605. doi: 10.3389/fphar.2019.00605. eCollection 2019. Front Pharmacol. 2019. PMID: 31244653 Free PMC article.
-
Quantification of receptor binding from response data obtained at different receptor levels: a simple individual sigmoid fitting and a unified SABRE approach.Sci Rep. 2022 Nov 6;12(1):18833. doi: 10.1038/s41598-022-23588-w. Sci Rep. 2022. PMID: 36336760 Free PMC article.
-
Reported Benefits of Low-Dose Naltrexone Appear to Be Independent of the Endogenous Opioid System Involving Proopiomelanocortin Neurons and β-Endorphin.eNeuro. 2021 Jun 16;8(3):ENEURO.0087-21.2021. doi: 10.1523/ENEURO.0087-21.2021. Print 2021 May-Jun. eNeuro. 2021. PMID: 34031099 Free PMC article.
-
μ-Opioid Receptor Activation Directly Modulates Intrinsically Photosensitive Retinal Ganglion Cells.Neuroscience. 2019 Jun 1;408:400-417. doi: 10.1016/j.neuroscience.2019.04.005. Epub 2019 Apr 11. Neuroscience. 2019. PMID: 30981862 Free PMC article.
-
Roux-en-Y gastric bypass in rat reduces mu-opioid receptor levels in brain regions associated with stress and energy regulation.PLoS One. 2019 Jun 20;14(6):e0218680. doi: 10.1371/journal.pone.0218680. eCollection 2019. PLoS One. 2019. PMID: 31220174 Free PMC article.
References
-
- Brown JH, Goldstein D (1986) Differences in muscarinic receptor reserve for inhibition of adenylate cyclase and stimulation of phosphoinositide hydrolysis in chick heart cells. Mol Pharmacol 30:566–570. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous
