Epac1-deficient mice have bleeding phenotype and thrombocytes with decreased GPIbβ expression
- PMID: 28821815
- PMCID: PMC5562764
- DOI: 10.1038/s41598-017-08975-y
Epac1-deficient mice have bleeding phenotype and thrombocytes with decreased GPIbβ expression
Abstract
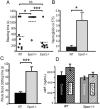
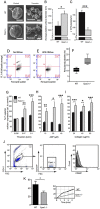
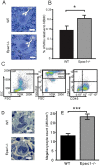
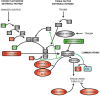
Epac1 (Exchange protein directly activated by cAMP 1) limits fluid loss from the circulation by tightening the endothelial barrier. We show here that Epac1-/- mice, but not Epac2-/- mice, have prolonged bleeding time, suggesting that Epac1 may limit fluid loss also by restraining bleeding. The Epac1-/- mice had deficient in vitro secondary hemostasis. Quantitative comprehensive proteomics analysis revealed that Epac1-/- mouse platelets (thrombocytes) had unbalanced expression of key components of the glycoprotein Ib-IX-V (GPIb-IX-V) complex, with decrease of GP1bβ and no change of GP1bα. This complex is critical for platelet adhesion under arterial shear conditions. Furthermore, Epac1-/- mice have reduced levels of plasma coagulation factors and fibrinogen, increased size of circulating platelets, increased megakaryocytes (the GP1bβ level was decreased also in Epac1-/- bone marrow) and higher abundance of reticulated platelets. Viscoelastic measurement of clotting function revealed Epac1-/- mice with a dysfunction in the clotting process, which corresponds to reduced plasma levels of coagulation factors like factor XIII and fibrinogen. We propose that the observed platelet phenotype is due to deficient Epac1 activity during megakaryopoiesis and thrombopoiesis, and that the defects in blood clotting for Epac1-/- is connected to secondary hemostasis.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

