G protein βγ subunits inhibit TRPM3 ion channels in sensory neurons
- PMID: 28826490
- PMCID: PMC5593501
- DOI: 10.7554/eLife.26138
G protein βγ subunits inhibit TRPM3 ion channels in sensory neurons
Abstract
Transient receptor potential (TRP) ion channels in peripheral sensory neurons are functionally regulated by hydrolysis of the phosphoinositide PI(4,5)P2 and changes in the level of protein kinase mediated phosphorylation following activation of various G protein coupled receptors. We now show that the activity of TRPM3 expressed in mouse dorsal root ganglion (DRG) neurons is inhibited by agonists of the Gi-coupled µ opioid, GABA-B and NPY receptors. These agonist effects are mediated by direct inhibition of TRPM3 by Gβγ subunits, rather than by a canonical cAMP mediated mechanism. The activity of TRPM3 in DRG neurons is also negatively modulated by tonic, constitutive GPCR activity as TRPM3 responses can be potentiated by GPCR inverse agonists. GPCR regulation of TRPM3 is also seen in vivo where Gi/o GPCRs agonists inhibited and inverse agonists potentiated TRPM3 mediated nociceptive behavioural responses.
Keywords: DRG; GPCR; TRP channels; ion channels; mouse; neuroscience; pain; sensory neuron.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures


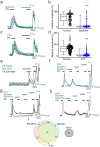
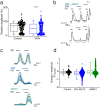
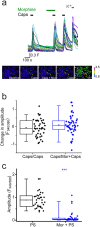

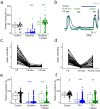

Comment in
-
A new target for G protein signaling.Elife. 2017 Sep 11;6:e31106. doi: 10.7554/eLife.31106. Elife. 2017. PMID: 28891794 Free PMC article.
References
-
- Agarwal N, Pacher P, Tegeder I, Amaya F, Constantin CE, Brenner GJ, Rubino T, Michalski CW, Marsicano G, Monory K, Mackie K, Marian C, Batkai S, Parolaro D, Fischer MJ, Reeh P, Kunos G, Kress M, Lutz B, Woolf CJ, Kuner R. Cannabinoids mediate analgesia largely via peripheral type 1 cannabinoid receptors in nociceptors. Nature Neuroscience. 2007;10:870–879. doi: 10.1038/nn1916. - DOI - PMC - PubMed
-
- Bardoni R, Tawfik VL, Wang D, François A, Solorzano C, Shuster SA, Choudhury P, Betelli C, Cassidy C, Smith K, de Nooij JC, Mennicken F, O'Donnell D, Kieffer BL, Woodbury CJ, Basbaum AI, MacDermott AB, Scherrer G. Delta opioid receptors presynaptically regulate cutaneous mechanosensory neuron input to the spinal cord dorsal horn. Neuron. 2014;81:1312–1327. doi: 10.1016/j.neuron.2014.01.044. - DOI - PMC - PubMed
-
- Bevan S, Quallo T, Andersson DA. TRPV1. In: Nilius B, Flockerzi V, editors. Handbook of Experimental Pharmacology. Berlin: Springer; 2014. pp. 207–245. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

