Improvement of Pharmacokinetic Profile of TRAIL via Trimer-Tag Enhances its Antitumor Activity in vivo
- PMID: 28827692
- PMCID: PMC5566391
- DOI: 10.1038/s41598-017-09518-1
Improvement of Pharmacokinetic Profile of TRAIL via Trimer-Tag Enhances its Antitumor Activity in vivo
Erratum in
-
Author Correction: Improvement of Pharmacokinetic Profile of TRAIL via Trimer-Tag Enhances its Antitumor Activity in vivo.Sci Rep. 2018 Mar 22;8(1):5266. doi: 10.1038/s41598-018-23057-3. Sci Rep. 2018. PMID: 29568007 Free PMC article.
Abstract
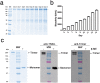
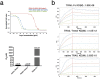
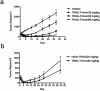
TNF-related apoptosis-inducing ligand (TRAIL/Apo2L) has long been considered a tantalizing target for cancer therapy because it mediates activation of the extrinsic apoptosis pathway in a tumor-specific manner by binding to and trimerizing its functional receptors DR4 or DR5. Despite initial promise, both recombinant human TRAIL (native TRAIL) and dimeric DR4/DR5 agonist monoclonal antibodies (mAbs) failed in multiple human clinical trials. Here we show that in-frame fusion of human C-propeptide of α1(I) collagen (Trimer-Tag) to the C-terminus of mature human TRAIL leads to a disulfide bond-linked homotrimer which can be expressed at high levels as a secreted protein from CHO cells. The resulting TRAIL-Trimer not only retains similar bioactivity and receptor binding kinetics as native TRAIL in vitro which are 4-5 orders of magnitude superior to that of dimeric TRAIL-Fc, but also manifests more favorable pharmacokinetic and antitumor pharmacodynamic profiles in vivo than that of native TRAIL. Taken together, this work provides direct evidence for the in vivo antitumor efficacy of TRAIL being proportional to systemic drug exposure and suggests that the previous clinical failures may have been due to rapid systemic clearance of native TRAIL and poor apoptosis-inducing potency of dimeric agonist mAbs despite their long serum half-lives.
Conflict of interest statement
P. Liang, X. Wang and J.G. Liang have ownership interest in Clover Biopharmaceuticals. P. Liang also has ownership interest in GenHunter Corporation. The other authors disclose no financial conflicts of interest.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

