Safety and Tolerability of Maraviroc-Containing Regimens to Prevent HIV Infection in Women: A Phase 2 Randomized Trial
- PMID: 28828489
- PMCID: PMC5667908
- DOI: 10.7326/M17-0520
Safety and Tolerability of Maraviroc-Containing Regimens to Prevent HIV Infection in Women: A Phase 2 Randomized Trial
Abstract
Background: Maraviroc (MVC) is a candidate drug for HIV preexposure prophylaxis (PrEP).
Objective: To assess the safety and tolerability of MVC-containing PrEP over 48 weeks in U.S. women at risk for HIV infection.
Design: Phase 2 randomized, controlled, double-blinded study of 4 antiretroviral regimens used as PrEP. (ClinicalTrials.gov: NCT01505114).
Setting: 12 clinical research sites of the HIV Prevention Trials Network and AIDS Clinical Trials Group.
Participants: HIV-uninfected women reporting condomless vaginal or anal intercourse with at least 1 man with HIV infection or unknown serostatus within 90 days.
Intervention: MVC only, MVC-emtricitabine (FTC), MVC-tenofovir disoproxil fumarate (TDF), and TDF-FTC (control).
Measurements: At each visit, clinical and laboratory (including HIV) assessments were done. Primary outcomes were grade 3 and 4 adverse events and time to permanent discontinuation of the study regimen. All randomly assigned participants were analyzed according to their original assignment.
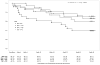
Results: Among 188 participants, 85% completed follow-up, 11% withdrew early, and 4% were lost to follow-up; 19% discontinued their regimen prematurely. The number discontinuing and the time to discontinuation did not differ among regimens. Grade 3 or 4 adverse events occurred in 5 (MVC), 13 (MVC-FTC), 9 (MVC-TDF), and 8 (TDF-FTC) participants; rates did not differ among regimens. One death (by suicide) occurred in the MVC-TDF group but was judged not to be related to study drugs. Of available plasma samples at week 48 (n = 126), 60% showed detectable drug concentrations. No new HIV infections occurred.
Limitations: Participants were not necessarily at high risk for HIV infection. The regimen comprised 3 pills taken daily. The study was not powered for efficacy.
Conclusion: Maraviroc-containing PrEP regimens were safe and well-tolerated compared with TDF-FTC in U.S. women. No new HIV infections occurred, although whether this was due to study drugs or low risk in the population is uncertain. Maraviroc-containing PrEP for women may warrant further study.
Primary funding source: National Institutes of Health.
Figures

References
-
- UNAIDS. [accessed 6/8/17];AIDS By The Numbers. 2016 Available at: http://www.unaids.org/sites/default/files/media_asset/AIDS-by-the-number....
-
- Thigpen MC, Kebaabetswe PM, Paxton LA, Smith DK, Rose CE, Segolodi TM, et al. Antiretroviral preexposure prophylaxis for heterosexual HIV transmission in Botswana. N Engl J Med. 2012;367:423–34. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
- UM1 AI069494/AI/NIAID NIH HHS/United States
- UM1 AI069423/AI/NIAID NIH HHS/United States
- UM1 AI069503/AI/NIAID NIH HHS/United States
- UM1 AI069501/AI/NIAID NIH HHS/United States
- U01 AI069423/AI/NIAID NIH HHS/United States
- UL1 TR001111/TR/NCATS NIH HHS/United States
- UL1 TR000005/TR/NCATS NIH HHS/United States
- UM1 AI069424/AI/NIAID NIH HHS/United States
- UL1 RR024153/RR/NCRR NIH HHS/United States
- UM1 AI069466/AI/NIAID NIH HHS/United States
- UL1 RR024996/RR/NCRR NIH HHS/United States
- UM1 AI069534/AI/NIAID NIH HHS/United States
- UM1 AI069415/AI/NIAID NIH HHS/United States
- P30 AI045008/AI/NIAID NIH HHS/United States
- UM1 AI069412/AI/NIAID NIH HHS/United States
- UM1 AI068634/AI/NIAID NIH HHS/United States
- UM1 AI069481/AI/NIAID NIH HHS/United States
- U54 GM104942/GM/NIGMS NIH HHS/United States
- P30 AI117970/AI/NIAID NIH HHS/United States
- UL1 TR002384/TR/NCATS NIH HHS/United States
- UM1 AI068619/AI/NIAID NIH HHS/United States
- UM1 AI106701/AI/NIAID NIH HHS/United States
- UM1 AI069465/AI/NIAID NIH HHS/United States
- UM1 AI068613/AI/NIAID NIH HHS/United States
- UM1 AI069419/AI/NIAID NIH HHS/United States
- U01 AI068634/AI/NIAID NIH HHS/United States
- UM1 AI068636/AI/NIAID NIH HHS/United States
- P30 AI050410/AI/NIAID NIH HHS/United States
- UM1 AI068617/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
