Cryopreservation and re-culture of a 2.3 litre biomass for use in a bioartificial liver device
- PMID: 28841674
- PMCID: PMC5572048
- DOI: 10.1371/journal.pone.0183385
Cryopreservation and re-culture of a 2.3 litre biomass for use in a bioartificial liver device
Abstract
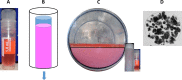
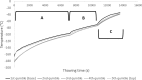
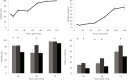
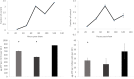
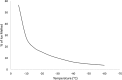
For large and complex tissue engineered constructs to be available on demand, long term storage using methods, such as cryopreservation, are essential. This study optimised parameters such as excess media concentration and warming rates and used the findings to enable the successful cryopreservation of 2.3 litres of alginate encapsulated liver cell spheroids. This volume of biomass is typical of those required for successful treatment of Acute Liver Failure using our Bioartificial Liver Device. Adding a buffer of medium above the biomass, as well as slow (0.6°C/min) warming rates was found to give the best results, so long as the warming through the equilibrium melting temperature was rapid. After 72 h post thaw-culture, viable cell number, glucose consumption, lactate production, and alpha-fetoprotein production had recovered to pre-freeze values in the 2.3 litre biomass (1.00 ± 0.05, 1.19 ± 0.10, 1.23 ± 0.18, 2.03 ± 0.04 per ml biomass of the pre-cryopreservation values respectively). It was also shown that further improvements in warming rates of the biomass could reduce recovery time to < 48 h. This is the first example of a biomass of this volume being successfully cryopreserved in a single cassette and re-cultured. It demonstrates that a bioartificial liver device can be cryopreserved, and has wider applications to scale-up large volume cryopreservation.
Conflict of interest statement
Figures
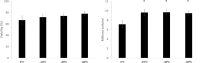
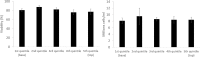

References
-
- Bernal W, Wendon J. Acute Liver Failure. New Engl J Med. 2013;369(26):2525–34. doi: 10.1056/NEJMra1208937 - DOI - PubMed
-
- Lee WM. Acute Liver Failure. Semin Resp Crit Care. 2012;33(1):36–45. doi: 10.1055/s-0032-1301733 - DOI - PubMed
-
- Nyberg SL, Mao SA, Glorioso JM. Bioartificial Liver In: Mitchell LMMN, editor. Pathobiology of Human Disease. San Diego: Academic Press; 2014. p. 1800–8.
-
- Erro E, Bundy J, Massie I, Chalmers SA, Gautier A, Gerontas S, et al. Bioengineering the liver: scale-up and cool chain delivery of the liver cell biomass for clinical targeting in a bioartificial liver support system. BioResearch open access. 2013;2(1):1–11. doi: 10.1089/biores.2012.0286 ; PubMed Central PMCID: PMC3569957. - DOI - PMC - PubMed
-
- Massie I, Selden C, Hodgson H, Fuller B. Cryopreservation of encapsulated liver spheroids for a bioartificial liver: reducing latent cryoinjury using an ice nucleating agent. Tissue engineering Part C, Methods. 2011;17(7):765–74. doi: 10.1089/ten.TEC.2010.0394 . - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

