Use of a Smartphone App to Assess Neonatal Jaundice
- PMID: 28842403
- PMCID: PMC5574723
- DOI: 10.1542/peds.2017-0312
Use of a Smartphone App to Assess Neonatal Jaundice
Abstract
Background: The assessment of jaundice in outpatient neonates is problematic. Visual assessment is inaccurate, and more exact methodologies are cumbersome and/or expensive. Our goal in this study was to assess the accuracy of a technology based on the analysis of digital images of newborns obtained using a smartphone application called BiliCam.
Methods: Paired BiliCam images and total serum bilirubin (TSB) levels were obtained in a diverse sample of newborns (<7 days old) at 7 sites across the United States. By using specialized software, data on color values in the images ("features") were extracted. Machine learning and regression analysis techniques were used to identify features for inclusion in models to predict an estimated bilirubin level for each newborn. The correlation between estimated bilirubin levels and TSB levels was calculated. In addition, the sensitivity and specificity of the estimated bilirubin levels in identifying newborns with high TSB levels were calculated by using 2 recommended decision rules for jaundice screening.
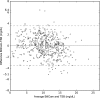
Results: Estimated bilirubin levels were calculated and compared with TSB levels in a diverse sample of 530 newborns (20.8% African American, 26.3% Hispanic, and 21.2% Asian American). The overall correlation was 0.91, and correlations among white, African American, Hispanic, and Asian American newborns were 0.92, 0.90, 0.91, and 0.88, respectively. The sensitivities of BiliCam in identifying newborns with high TSB levels were 84.6% and 100%, respectively, by using 2 decision rules; specificities were 75.1% and 76.4%, respectively.
Conclusions: BiliCam provided accurate estimates of TSB values, demonstrating that an inexpensive technology that uses commodity smartphones could be used to effectively screen newborns for jaundice.
Copyright © 2017 by the American Academy of Pediatrics.
Conflict of interest statement
POTENTIAL CONFLICT OF INTEREST: Drs Taylor, Stout, and Patel are cofounders of BiliCam, LLC, a company developing the technology described in this study for commercial use; the other authors have indicated they have no potential conflicts of interest to disclose.
Figures

Comment in
-
Phone app detects hyperbilirubinemia.J Pediatr. 2018 Mar;194:265-268. doi: 10.1016/j.jpeds.2017.12.078. J Pediatr. 2018. PMID: 29478501 No abstract available.
References
-
- Bhutani VK, Stark AR, Lazzeroni LC, et al. Predischarge screening for severe neonatal hyperbilirubinemia identifies infants who need phototherapy. J Pediatr. 2013;162(3):477–482.e1 - PubMed
-
- Keren R, Tremont K, Luan X, Cnaan A. Visual assessment of jaundice in term and late preterm infants. Arch Dis Child Fetal Neonatal Ed. 2009;94(5):F317–F322 - PubMed
-
- Maisels MJ, Bhutani VK, Bogen D, Newman TB, Stark AR, Watchko JF. Hyperbilirubinemia in the newborn infant > or =35 weeks’ gestation: an update with clarifications. Pediatrics. 2009;124(4):1193–1198 - PubMed
-
- Datar A, Sood N. Impact of postpartum hospital-stay legislation on newborn length of stay, readmission, and mortality in California. Pediatrics. 2006;118(1):63–72 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

