The Atypical MAP Kinase SWIP-13/ERK8 Regulates Dopamine Transporters through a Rho-Dependent Mechanism
- PMID: 28842414
- PMCID: PMC5607470
- DOI: 10.1523/JNEUROSCI.1582-17.2017
The Atypical MAP Kinase SWIP-13/ERK8 Regulates Dopamine Transporters through a Rho-Dependent Mechanism
Abstract
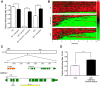
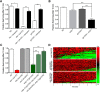
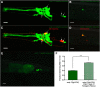
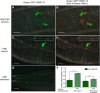
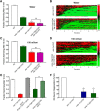
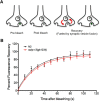
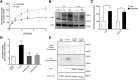
The neurotransmitter dopamine (DA) regulates multiple behaviors across phylogeny, with disrupted DA signaling in humans associated with addiction, attention-deficit/ hyperactivity disorder, schizophrenia, and Parkinson's disease. The DA transporter (DAT) imposes spatial and temporal limits on DA action, and provides for presynaptic DA recycling to replenish neurotransmitter pools. Molecular mechanisms that regulate DAT expression, trafficking, and function, particularly in vivo, remain poorly understood, though recent studies have implicated rho-linked pathways in psychostimulant action. To identify genes that dictate the ability of DAT to sustain normal levels of DA clearance, we pursued a forward genetic screen in Caenorhabditis elegans based on the phenotype swimming-induced paralysis (Swip), a paralytic behavior observed in hermaphrodite worms with loss-of-function dat-1 mutations. Here, we report the identity of swip-13, which encodes a highly conserved ortholog of the human atypical MAP kinase ERK8. We present evidence that SWIP-13 acts presynaptically to insure adequate levels of surface DAT expression and DA clearance. Moreover, we provide in vitro and in vivo evidence supporting a conserved pathway involving SWIP-13/ERK8 activation of Rho GTPases that dictates DAT surface expression and function.SIGNIFICANCE STATEMENT Signaling by the neurotransmitter dopamine (DA) is tightly regulated by the DA transporter (DAT), insuring efficient DA clearance after release. Molecular networks that regulate DAT are poorly understood, particularly in vivo Using a forward genetic screen in the nematode Caenorhabditis elegans, we implicate the atypical mitogen activated protein kinase, SWIP-13, in DAT regulation. Moreover, we provide in vitro and in vivo evidence that SWIP-13, as well as its human counterpart ERK8, regulate DAT surface availability via the activation of Rho proteins. Our findings implicate a novel pathway that regulates DA synaptic availability and that may contribute to risk for disorders linked to perturbed DA signaling. Targeting this pathway may be of value in the development of therapeutics in such disorders.
Keywords: C. elegans; dopamine; genetics; kinase; neurotransmitters; transporters.
Copyright © 2017 the authors 0270-6474/17/379288-17$15.00/0.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
- T32 MH064913/MH/NIMH NIH HHS/United States
- R01 NS078291/NS/NINDS NIH HHS/United States
- F31 DA035559/DA/NIDA NIH HHS/United States
- P30 DK058404/DK/NIDDK NIH HHS/United States
- F31 MH093102/MH/NIMH NIH HHS/United States
- U2C DK059637/DK/NIDDK NIH HHS/United States
- U54 HD083211/HD/NICHD NIH HHS/United States
- P30 EY008126/EY/NEI NIH HHS/United States
- P30 DK020593/DK/NIDDK NIH HHS/United States
- P30 CA068485/CA/NCI NIH HHS/United States
- F31 MH109196/MH/NIMH NIH HHS/United States
- R01 MH095044/MH/NIMH NIH HHS/United States
- U24 DK059637/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
