The activin receptor is stimulated in the skeleton, vasculature, heart, and kidney during chronic kidney disease
- PMID: 28843411
- PMCID: PMC6628245
- DOI: 10.1016/j.kint.2017.06.016
The activin receptor is stimulated in the skeleton, vasculature, heart, and kidney during chronic kidney disease
Abstract
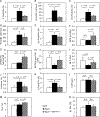
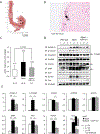
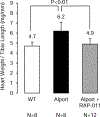
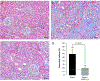
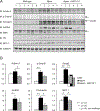
We examined activin receptor type IIA (ActRIIA) activation in chronic kidney disease (CKD) by signal analysis and inhibition in mice with Alport syndrome using the ActRIIA ligand trap RAP-011 initiated in 75-day-old Alport mice. At 200 days of age, there was severe CKD and associated Mineral and Bone Disorder (CKD-MBD), consisting of osteodystrophy, vascular calcification, cardiac hypertrophy, hyperphosphatemia, hyperparathyroidism, elevated FGF23, and reduced klotho. The CKD-induced bone resorption and osteoblast dysfunction was reversed, and bone formation was increased by RAP-011. ActRIIA inhibition prevented the formation of calcium apatite deposits in the aortic adventitia and tunica media and significantly decreased the mean aortic calcium concentration from 0.59 in untreated to 0.36 mg/g in treated Alport mice. Aortic ActRIIA stimulation in untreated mice increased p-Smad2 levels and the transcription of sm22α and αSMA. ActRIIA inhibition reversed aortic expression of the osteoblast transition markers Runx2 and osterix. Heart weight was significantly increased by 26% in untreated mice but remained normal during RAP-011 treatment. In 150-day-old mice, GFR was significantly reduced by 55%, but only by 30% in the RAP-011-treated group. In 200-day-old mice, the mean BUN was 100 mg/dl in untreated mice compared to 60 mg/dl in the treated group. In the kidneys of 200-day-old mice, ActRIIA and p-Smad2 were induced and MCP-1, fibronectin, and interstitial fibrosis were stimulated; all were attenuated by RAP-011 treatment. Hence, the activation of ActRIIA signaling during early CKD contributes to the CKD-MBD components of osteodystrophy and cardiovascular disease and to renal fibrosis. Thus, the inhibition of ActRIIA signaling is efficacious in improving and delaying CKD-MBD in this model of Alport syndrome.
Keywords: bone; cardiovascular disease; chronic kidney disease; fibrosis; mineral metabolism; vascular calcification.
Copyright © 2017 International Society of Nephrology. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Disclosure:
KAH and HHM have been Celgene consultants. The other authors have no competing financial interests.
Figures
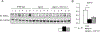
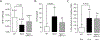
References
-
- Block GA, Hulbert-Shearon TE, Levin NW, Port FK. Association of serum phosphorus and calcium X phosphate product with mortality risk in chronic hemodialysis patients: a national study. Am J Kidney Dis 1998;31(4):607–17. - PubMed
-
- Go AS, Chertow GM, Fan D, McCulloch CE, Hsu Cy. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. New Engl J Med 2004;351:1296–305. - PubMed
-
- Blacher J, Guerin AP, Pannier B, Marchais SJ, London GM. Arterial calcifications, arterial stiffness, and cardiovascular risk in end-stage renal disease. Hypertension 2001;38:938–42. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

