ESCRT-dependent control of membrane remodelling during cell division
- PMID: 28843980
- PMCID: PMC6015221
- DOI: 10.1016/j.semcdb.2017.08.035
ESCRT-dependent control of membrane remodelling during cell division
Abstract
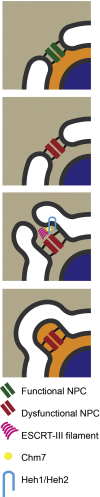
The Endosomal Sorting Complex Required for Transport (ESCRT) proteins form an evolutionarily conserved membrane remodelling machinery. Identified originally for their role in cargo sorting and remodelling of endosomal membranes during yeast vacuolar sorting, an extensive body of work now implicates a sub-complex of this machinery (ESCRT-III), as a transplantable membrane fission machinery that is dispatched to various cellular locations to achieve a topologically unique membrane separation. Surprisingly, several ESCRT-III-regulated processes occur during cell division, when cells undergo a dramatic and co-ordinated remodelling of their membranes to allow the physical processes of division to occur. The ESCRT machinery functions in regeneration of the nuclear envelope during open mitosis and in the abscission phase of cytokinesis, where daughter cells are separated from each other in the last act of division. Roles for the ESCRT machinery in cell division are conserved as far back as Archaea, suggesting that the ancestral role of these proteins was as a membrane remodelling machinery that facilitated division and that was co-opted throughout evolution to perform a variety of other cell biological functions. Here, we will explore the function and regulation of the ESCRT machinery in cell division.
Keywords: Abscission; Cytokinesis; ESCRT; Endosomal sorting complex required for transport; Nuclear envelope.
Copyright © 2017 The Authors. Published by Elsevier Ltd.. All rights reserved.
Figures
Similar articles
-
The ESCRT-machinery: closing holes and expanding roles.Curr Opin Cell Biol. 2019 Aug;59:121-132. doi: 10.1016/j.ceb.2019.04.005. Epub 2019 May 24. Curr Opin Cell Biol. 2019. PMID: 31132588 Review.
-
Regulation of cytokinesis by membrane trafficking involving small GTPases and the ESCRT machinery.Crit Rev Biochem Mol Biol. 2016;51(1):1-6. doi: 10.3109/10409238.2015.1085827. Epub 2015 Sep 11. Crit Rev Biochem Mol Biol. 2016. PMID: 26362026 Review.
-
A relay race of ESCRT-III paralogs drives cell division in a hyperthermophilic archaeon.mBio. 2025 Feb 5;16(2):e0099124. doi: 10.1128/mbio.00991-24. Epub 2024 Dec 19. mBio. 2025. PMID: 39699168 Free PMC article.
-
A Septin Double Ring Controls the Spatiotemporal Organization of the ESCRT Machinery in Cytokinetic Abscission.Curr Biol. 2019 Jul 8;29(13):2174-2182.e7. doi: 10.1016/j.cub.2019.05.050. Epub 2019 Jun 13. Curr Biol. 2019. PMID: 31204162 Free PMC article.
-
Dynamics of endosomal sorting complex required for transport (ESCRT) machinery during cytokinesis and its role in abscission.Proc Natl Acad Sci U S A. 2011 Mar 22;108(12):4846-51. doi: 10.1073/pnas.1102714108. Epub 2011 Mar 7. Proc Natl Acad Sci U S A. 2011. PMID: 21383202 Free PMC article.
Cited by
-
Extrachromosomal Histone H2B Contributes to the Formation of the Abscission Site for Cell Division.Cells. 2019 Nov 5;8(11):1391. doi: 10.3390/cells8111391. Cells. 2019. PMID: 31694230 Free PMC article.
-
CHMP2A regulates broad immune cell-mediated antitumor activity in an immunocompetent in vivo head and neck squamous cell carcinoma model.J Immunother Cancer. 2024 May 3;12(5):e007187. doi: 10.1136/jitc-2023-007187. J Immunother Cancer. 2024. PMID: 38702144 Free PMC article.
-
Induction of pyroptotic cell death as a potential tool for cancer treatment.J Inflamm (Lond). 2022 Nov 14;19(1):19. doi: 10.1186/s12950-022-00316-9. J Inflamm (Lond). 2022. PMID: 36376979 Free PMC article. Review.
-
Phase Separation in the Nucleus and at the Nuclear Periphery during Post-Mitotic Nuclear Envelope Reformation.Cells. 2022 May 25;11(11):1749. doi: 10.3390/cells11111749. Cells. 2022. PMID: 35681444 Free PMC article. Review.
-
The Flemmingsome reveals an ESCRT-to-membrane coupling via ALIX/syntenin/syndecan-4 required for completion of cytokinesis.Nat Commun. 2020 Apr 22;11(1):1941. doi: 10.1038/s41467-020-15205-z. Nat Commun. 2020. PMID: 32321914 Free PMC article.
References
-
- Katzmann D.J., Babst M., Emr S.D. Ubiquitin-dependent sorting into the multivesicular body pathway requires the function of a conserved endosomal protein sorting complex, ESCRT-I. Cell. 2001;106:145–155. - PubMed
-
- Babst M., Katzmann D.J., Snyder W.B., Wendland B., Emr S.D. Endosome-associated complex ESCRT-II, recruits transport machinery for protein sorting at the multivesicular body. Dev. Cell. 2002;3:283–289. - PubMed
-
- Babst M., Katzmann D.J., Estepa-Sabal E.J., Meerloo T., Emr S.D. Escrt-III: an endosome-associated heterooligomeric protein complex required for mvb sorting. Dev. Cell. 2002;3:271–282. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

