In Vivo Magnetic Recording of Neuronal Activity
- PMID: 28844526
- PMCID: PMC5744593
- DOI: 10.1016/j.neuron.2017.08.012
In Vivo Magnetic Recording of Neuronal Activity
Abstract
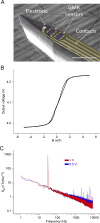
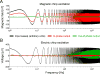
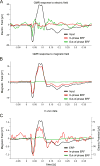
Neuronal activity generates ionic flows and thereby both magnetic fields and electric potential differences, i.e., voltages. Voltage measurements are widely used but suffer from isolating and smearing properties of tissue between source and sensor, are blind to ionic flow direction, and reflect the difference between two electrodes, complicating interpretation. Magnetic field measurements could overcome these limitations but have been essentially limited to magnetoencephalography (MEG), using centimeter-sized, helium-cooled extracranial sensors. Here, we report on in vivo magnetic recordings of neuronal activity from visual cortex of cats with magnetrodes, specially developed needle-shaped probes carrying micron-sized, non-cooled magnetic sensors based on spin electronics. Event-related magnetic fields inside the neuropil were on the order of several nanoteslas, informing MEG source models and efforts for magnetic field measurements through MRI. Though the signal-to-noise ratio is still inferior to electrophysiology, this proof of concept demonstrates the potential to exploit the fundamental advantages of magnetophysiology.
Keywords: MEG; Magnetic fields; magnetic sensors; magnetoencephalography; spin electronics.
Copyright © 2017 Elsevier Inc. All rights reserved.
Figures



References
-
- Amaral J, Cardoso S, Freitas PP, Sebastião AM. Toward a system to measure action potential on mice brain slices with local magnetoresistive probes. Journal of Applied Physics. 2011;109:07B308.
-
- Baibich MN, Broto JM, Fert A, Nguyen Van Dau F, Petroff F, Etienne P, Creuzet G, Friederich A, Chazelas J. Giant magnetoresistance of (001)Fe/(001)Cr magnetic superlattices. Physical review letters. 1988;61:2472–2475. - PubMed
-
- Bandettini PA, Petridou N, Bodurka J. Direct detection of neuronal activity with MRI: Fantasy, possibility, or reality? Applied Magnetic Resonance. 2005;29:65–88.
-
- Barnes FS, Greenebaum B. Biological and medical aspects of electromagnetic fields. 3. CRC Press; 2007. c effects of electromagnetic fields.
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

