The dynamics of the metabolism of acetate and bicarbonate associated with use of hemodialysates in the ABChD trial: a phase IV, prospective, single center, single blind, randomized, cross-over, two week investigation
- PMID: 28851317
- PMCID: PMC5576126
- DOI: 10.1186/s12882-017-0683-6
The dynamics of the metabolism of acetate and bicarbonate associated with use of hemodialysates in the ABChD trial: a phase IV, prospective, single center, single blind, randomized, cross-over, two week investigation
Abstract
Background: In the United States, hemodialysis (HD) is generally performed via a bicarbonate dialysate. It is not known if small amounts of acid used in dialysate to buffer the bicarbonate can meaningfully contribute to overall buffering administered during HD. We aimed to investigate the metabolism of acetate with use of two different acid buffer concentrates and determine if it effects blood bicarbonate concentrations in HD patients.
Methods: The Acid-Base Composition with use of hemoDialysates (ABChD) trial was a Phase IV, prospective, single blind, randomized, cross-over, 2 week investigation of peridialytic dynamics of acetate and bicarbonate associated with use of acid buffer concentrates. Eleven prevalent HD patients participated from November 2014 to February 2015. Patients received two HD treatments, with NaturaLyte® and GranuFlo® acid concentrates containing 4 and 8 mEq/L of acetate, respectively. Dialysate order was chosen in a random fashion. The endpoint was to characterize the dynamics of acetate received and metabolized during hemodialysis, and how it effects overall bicarbonate concentrations in the blood and dialysate. Acetate and bicarbonate concentrations were assessed before, at 8 time points during, and 6 time points after the completion of HD.
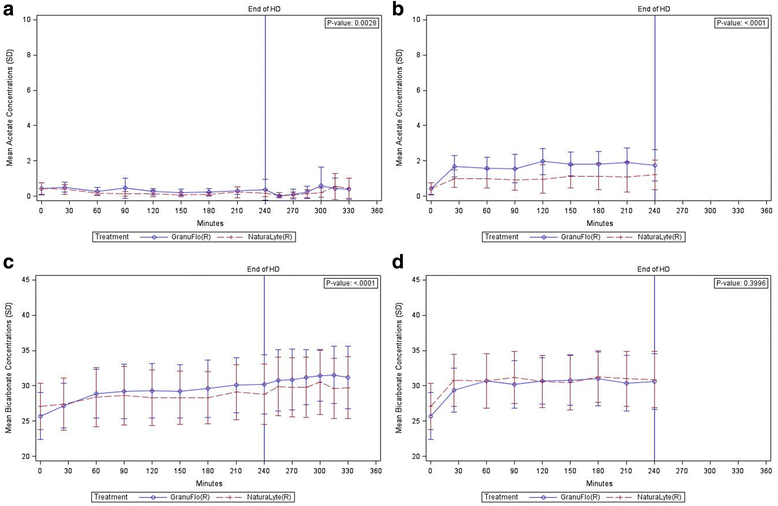
Results: Data from 20 HD treatments for 11 patients (10 NaturaLyte® and 10 GranuFlo®) was analyzed. Cumulative trajectories of arterialized acetate were unique between NaturaLyte® and GranuFlo® (p = 0.003), yet individual time points demonstrated overlap without remarkable differences. Arterialized and venous blood bicarbonate concentrations were similar at HD initiation, but by 240 min into dialysis, mean arterialized bicarbonate concentrations were 30.2 (SD ± 4.16) mEq/L in GranuFlo® and 28.8 (SD ± 4.26) mEq/L in NaturaLyte®. Regardless of acid buffer concentrate, arterial blood bicarbonate was primarily dictated by the prescribed bicarbonate level. Subjects tolerated HD with both acid buffer concentrates without experiencing any related adverse events.
Conclusions: A small fraction of acetate was delivered to HD patients with use of NaturaLyte® and GranuFlo® acid buffers; the majority of acetate received was observed to be rapidly metabolized and cleared from the circulation. Blood bicarbonate concentrations appear to be determined mainly by the prescribed concentration of bicarbonate.
Trial registration: This trial was registered on ClinicalTrials.gov on 11 Dec 2014 ( NCT02334267 ).
Keywords: Acetate; Acid-Base; Bicarbonate; Dialysate; Hemodialysis; Hemodynamics; Metabolism.
Conflict of interest statement
Ethics approval and consent to participate
This study was conducted under a protocol approved by Crescent City Institutional Review Board (IRB) located at 2820 Canal Street, New Orleans, LA 70119, and all patients were provided informed consent prior to study participation (IRB Protocol # 14–034).
Consent for publication
Not applicable.
Competing interests
The hemodialysis acid concentrate buffers used in this clinical trial are patented commercial medical devices of Fresenius Medical Care North America. William B. Smith and Sandy Gibson: Employed by Volunteer Research Group and New Orleans Center for Clinical Research at the University of Tennessee Medical Center. James Zhao and Margarita Askelson: Employed by EDETEK. Jamil Hantash: Employed by Tandem Labs. Brigid Flanagan: Employed by Frenova Renal Research; Stock ownership in Fresenius Medical Care. George E. Newman, and Kendra S. Hendon: Associates/physicians of Knoxville Kidney Center, PLLC. John W. Larkin: Employed by Fresenius Medical Care North America. Len A. Usvyat: Employed by Fresenius Medical Care North America; Stock ownership in Fresenius Medical Care. Ravi I Thadhani: Employed by Massachusetts General Hospital and Partners Healthcare; Consultancy Agreements: Biogen Idec; Independent research consultant of Fresenius Medical Care North America, Pfizer, Shire Human Genetics, and Celgene; The costs of travel and accommodations related to this clinical trial were reimbursed by Fresenius Medical Care North America; Ownership interests in Aggamin LLC; Receive research funding from the National Institutes of Health, Kaneka Corporation, Diasorin, and ThermoFisher; Receive honoraria from Roche Diagnostics, ZS Pharma; Patents and invention interests include ThermoFisher and Up-To-Date; Scientific advisor for Aggamin LLC. Franklin W. Maddux: Employed by Fresenius Medical Care North America; Stock ownership in Fresenius Medical Care; Directorships in American National Bank & Trust (NASDAQ: AMNB), Mid-Atlantic Renal Coalition, Specialty Care, Inc., and Sound Physicians; Chairman Pacific Care Renal Foundation 501c3 nonprofit; Chair - Kidney Care Partners; Founder - Scholarships Expanding Education 501c3 nonprofit.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
-
- National Kidney F. K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis. 2003;42(4 Suppl 3):S1–201. - PubMed
-
- Lowrie EG, Lew NL. Commonly measured laboratory variables in hemodialysis patients: relationships among them and to death risk. Semin Nephrol. 1992;12(3):276–283. - PubMed
-
- Bommer J, Locatelli F, Satayathum S, Keen ML, Goodkin DA, Saito A, Akiba T, Port FK, Young EW. Association of predialysis serum bicarbonate levels with risk of mortality and hospitalization in the dialysis outcomes and practice patterns study (DOPPS) Am J Kidney Dis. 2004;44(4):661–671. doi: 10.1016/S0272-6386(04)00936-9. - DOI - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

