Therapeutic effect of human adipose-derived stem cells and their secretome in experimental diabetic pain
- PMID: 28851944
- PMCID: PMC5575274
- DOI: 10.1038/s41598-017-09487-5
Therapeutic effect of human adipose-derived stem cells and their secretome in experimental diabetic pain
Abstract
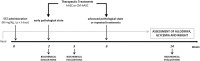
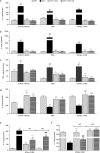
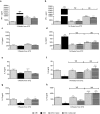
Painful neuropathy is one of the complications of diabetes mellitus that adversely affects patients'quality of life. Pharmacological treatments are not fully satisfactory, and novel approaches needed. In a preclinical mouse model of diabetes the effect of both human mesenchymal stromal cells from adipose tissue (hASC) and their conditioned medium (hASC-CM) was evaluated. Diabetes was induced by streptozotocin. After neuropathic hypersensitivity was established, mice were intravenously injected with either 1 × 106 hASC or with CM derived from 2 × 106 hASC. Both hASC and CM (secretome) reversed mechanical, thermal allodynia and thermal hyperalgesia, with a rapid and long lasting effect, maintained up to 12 weeks after treatments. In nerves, dorsal root ganglia and spinal cord of neuropathic mice we determined high IL-1β, IL-6 and TNF-α and low IL-10 levels. Both treatments restored a correct pro/antinflammatory cytokine balance and prevented skin innervation loss. In spleens of streptozotocin-mice, both hASC and hASC-CM re-established Th1/Th2 balance that was shifted to Th1 during diabetes. Blood glucose levels were unaffected although diabetic animals regained weight, and kidney morphology was recovered by treatments. Our data show that hASC and hASC-CM treatments may be promising approaches for diabetic neuropathic pain, and suggest that cell effect is likely mediated by their secretome.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures




Similar articles
-
Secretome of human adipose-derived mesenchymal stem cell relieves pain and neuroinflammation independently of the route of administration in experimental osteoarthritis.Brain Behav Immun. 2021 May;94:29-40. doi: 10.1016/j.bbi.2021.03.011. Epub 2021 Mar 15. Brain Behav Immun. 2021. PMID: 33737173
-
Bone marrow-derived mesenchymal stem/stromal cells reverse the sensorial diabetic neuropathy via modulation of spinal neuroinflammatory cascades.J Neuroinflammation. 2018 Jun 22;15(1):189. doi: 10.1186/s12974-018-1224-3. J Neuroinflammation. 2018. PMID: 29933760 Free PMC article.
-
Effects of Transplantation of CTLA4Ig-Overexpressing Adipose Tissue-Derived Mesenchymal Stem Cells in Mice With Sustained Severe Rheumatoid Arthritis.Cell Transplant. 2016;25(2):243-59. doi: 10.3727/096368915X688470. Epub 2015 Jun 17. Cell Transplant. 2016. PMID: 26087385
-
Human Adipose-Derived Stromal/Stem Cell Culture and Analysis Methods for Adipose Tissue Modeling In Vitro: A Systematic Review.Cells. 2021 Jun 3;10(6):1378. doi: 10.3390/cells10061378. Cells. 2021. PMID: 34204869 Free PMC article.
-
Factors Defining Human Adipose Stem/Stromal Cell Immunomodulation in Vitro.Stem Cell Rev Rep. 2024 Jan;20(1):175-205. doi: 10.1007/s12015-023-10654-7. Epub 2023 Nov 14. Stem Cell Rev Rep. 2024. PMID: 37962697 Free PMC article. Review.
Cited by
-
Effect of adipose-derived mesenchymal stem cells on hepatocellular carcinoma: In vitro inhibition of carcinogenesis.World J Gastroenterol. 2019 Feb 7;25(5):567-583. doi: 10.3748/wjg.v25.i5.567. World J Gastroenterol. 2019. PMID: 30774272 Free PMC article.
-
All the PNS is a Stage: Transplanted Bone Marrow Cells Play an Immunomodulatory Role in Peripheral Nerve Regeneration.ASN Neuro. 2023 Jan-Dec;15:17590914231167281. doi: 10.1177/17590914231167281. ASN Neuro. 2023. PMID: 37654230 Free PMC article.
-
Role of adipose mesenchymal stem cells and secretome in peripheral nerve regeneration.Ann Med Surg (Lond). 2021 Jun 9;67:102482. doi: 10.1016/j.amsu.2021.102482. eCollection 2021 Jul. Ann Med Surg (Lond). 2021. PMID: 34168873 Free PMC article. Review.
-
The Future of Regenerative Medicine: Cell Therapy Using Pluripotent Stem Cells and Acellular Therapies Based on Extracellular Vesicles.Cells. 2021 Jan 27;10(2):240. doi: 10.3390/cells10020240. Cells. 2021. PMID: 33513719 Free PMC article. Review.
-
Reestablishment of Redox Homeostasis in the Nociceptive Primary Afferent as a Mechanism of Antinociception Promoted by Mesenchymal Stem/Stromal Cells in Oxaliplatin-Induced Chronic Peripheral Neuropathy.Stem Cells Int. 2021 Jan 6;2021:8815206. doi: 10.1155/2021/8815206. eCollection 2021. Stem Cells Int. 2021. PMID: 33505472 Free PMC article.
References
-
- Aring AM, Jones DE, Falko JM. Evaluation and prevention of diabetic neuropathy. Am Fam Physician. 2005;71:2123–2128. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

