Mechanisms of stem cell based cardiac repair-gap junctional signaling promotes the cardiac lineage specification of mesenchymal stem cells
- PMID: 28852100
- PMCID: PMC5574972
- DOI: 10.1038/s41598-017-10122-6
Mechanisms of stem cell based cardiac repair-gap junctional signaling promotes the cardiac lineage specification of mesenchymal stem cells
Abstract
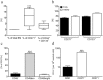
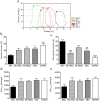
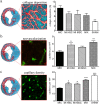
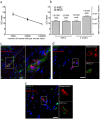
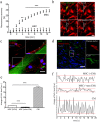
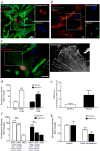
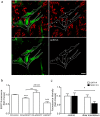
Different subtypes of bone marrow-derived stem cells are characterized by varying functionality and activity after transplantation into the infarcted heart. Improvement of stem cell therapeutics requires deep knowledge about the mechanisms that mediate the benefits of stem cell treatment. Here, we demonstrated that co-transplantation of mesenchymal stem cells (MSCs) and hematopoietic stem cells (HSCs) led to enhanced synergistic effects on cardiac remodeling. While HSCs were associated with blood vessel formation, MSCs were found to possess transdifferentiation capacity. This cardiomyogenic plasticity of MSCs was strongly promoted by a gap junction-dependent crosstalk between myocytes and stem cells. The inhibition of cell-cell coupling significantly reduced the expression of the cardiac specific transcription factors NKX2.5 and GATA4. Interestingly, we observed that small non-coding RNAs are exchanged between MSCs and cardiomyocytes in a GJ-dependent manner that might contribute to the transdifferentiation process of MSCs within a cardiac environment. Our results suggest that the predominant mechanism of HSCs contribution to cardiac regeneration is based on their ability to regulate angiogenesis. In contrast, transplanted MSCs have the capability for intercellular communication with surrounding cardiomyocytes, which triggers the intrinsic program of cardiogenic lineage specification of MSCs by providing cardiomyocyte-derived cues.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
Pre-treatment of mesenchymal stem cells with a combination of growth factors enhances gap junction formation, cytoprotective effect on cardiomyocytes, and therapeutic efficacy for myocardial infarction.J Am Coll Cardiol. 2008 Mar 4;51(9):933-43. doi: 10.1016/j.jacc.2007.11.040. J Am Coll Cardiol. 2008. PMID: 18308163 Clinical Trial.
-
Pretreatment of mesenchymal stem cells with angiotensin II enhances paracrine effects, angiogenesis, gap junction formation and therapeutic efficacy for myocardial infarction.Int J Cardiol. 2015 Jun 1;188:22-32. doi: 10.1016/j.ijcard.2015.03.425. Epub 2015 Apr 1. Int J Cardiol. 2015. PMID: 25880576
-
Mechanistic effects of mesenchymal and hematopoietic stem cells: New therapeutic targets in myocardial infarction.J Cell Biochem. 2018 Jul;119(7):5274-5286. doi: 10.1002/jcb.26637. Epub 2018 Mar 12. J Cell Biochem. 2018. PMID: 29266431 Review.
-
Preinduction with bone morphogenetic protein-2 enhances cardiomyogenic differentiation of c-kit+ mesenchymal stem cells and repair of infarcted myocardium.Int J Cardiol. 2018 Aug 15;265:173-180. doi: 10.1016/j.ijcard.2018.01.134. Int J Cardiol. 2018. PMID: 29885685
-
Mesenchymal stem cell therapy: A promising cell-based therapy for treatment of myocardial infarction.J Gene Med. 2017 Dec;19(12). doi: 10.1002/jgm.2995. Epub 2017 Dec 8. J Gene Med. 2017. PMID: 29044850 Review.
Cited by
-
Dose-Independent Therapeutic Benefit of Bone Marrow Stem Cell Transplantation after MI in Mice.Biomedicines. 2020 Jun 11;8(6):157. doi: 10.3390/biomedicines8060157. Biomedicines. 2020. PMID: 32545336 Free PMC article.
-
Biomaterials Loaded with Growth Factors/Cytokines and Stem Cells for Cardiac Tissue Regeneration.Int J Mol Sci. 2020 Aug 19;21(17):5952. doi: 10.3390/ijms21175952. Int J Mol Sci. 2020. PMID: 32824966 Free PMC article. Review.
-
Intramyocardial angiogenetic stem cells and epicardial erythropoietin save the acute ischemic heart.Dis Model Mech. 2018 Jun 22;11(6):dmm033282. doi: 10.1242/dmm.033282. Dis Model Mech. 2018. PMID: 29752300 Free PMC article.
-
Angiogenic Potential of Bone Marrow Derived CD133+ and CD271+ Intramyocardial Stem Cell Trans- Plantation Post MI.Cells. 2019 Dec 27;9(1):78. doi: 10.3390/cells9010078. Cells. 2019. PMID: 31892273 Free PMC article.
-
The Impact of Spaceflight and Microgravity on the Human Islet-1+ Cardiovascular Progenitor Cell Transcriptome.Int J Mol Sci. 2021 Mar 30;22(7):3577. doi: 10.3390/ijms22073577. Int J Mol Sci. 2021. PMID: 33808224 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

