Identification of WEE1 as a target to make AKT inhibition more effective in melanoma
- PMID: 28853983
- PMCID: PMC5790369
- DOI: 10.1080/15384047.2017.1360446
Identification of WEE1 as a target to make AKT inhibition more effective in melanoma
Abstract
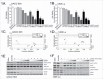
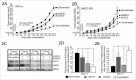
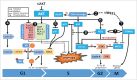
AKT3 is one of the major therapeutic targets in melanoma but clinically targeting AKT3 alone seems to be an ineffective therapeutic approach. To identify unique strategies to enhance the efficacy of targeting AKT3, a screen was undertaken where AKT3 was co-targeted with a panel of kinases important in melanoma development. The screen identified WEE1 as the most potent target that when inhibited along with AKT3 would enhance the efficacy of targeting AKT3 in melanoma. RNAi mediated inhibition of AKT3 and WEE1 synergistically inhibited the viability of melanoma cells leading to a 65-75% decrease in tumor development. This approach was effective by mechanistically modulating pathways associated with the transcription factors p53 and FOXM1. Simultaneously regulating the activity of these two transcriptionally driven pathways, cooperatively deregulated cell cycle control and DNA damage repair to synergistically kill melanoma cells. This study uniquely identifies a potential approach to improve the efficacy of targeting AKT3 in melanoma.
Keywords: AKT3; Melanoma; WEE1; synergy.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
