Cancer drug funding decisions in Scotland: impact of new end-of-life, orphan and ultra-orphan processes
- PMID: 28854927
- PMCID: PMC5577765
- DOI: 10.1186/s12913-017-2561-0
Cancer drug funding decisions in Scotland: impact of new end-of-life, orphan and ultra-orphan processes
Abstract
Background: The Scottish Medicines Consortium evaluates new drugs for use in the National Health Service in Scotland. Reforms in 2014 to their evaluation process aimed to increase patient access to new drugs for end-of-life or rare conditions; the changes include additional steps in the process to gain further information from patients and clinicians, and for revised commercial agreements. This study examines the extent of any impact of the reforms on funding decisions.
Method: Data on the Scottish Medicines Consortium's funding decisions during 24 months post-reform were extracted from published Advice, for descriptive statistics and thematic analysis. Comparison data were extracted for the 24 months pre-reform. Data on decisions for England by the National Institute for Clinical and Health Excellence for the same drugs were extracted from published Technology Appraisals.
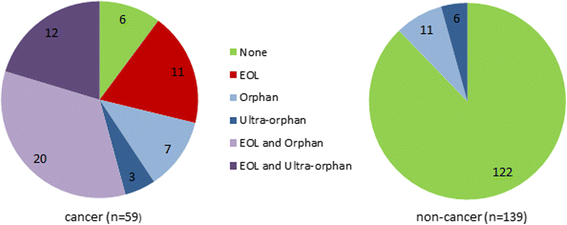
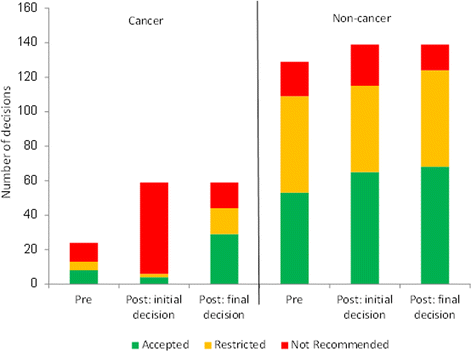
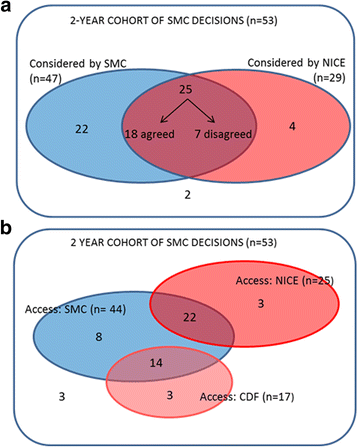
Results: The new process was used by 90% (53/59) of cancer submissions. It is triggered if the initial advice is not to recommend, and this risk-of-rejection level is higher than in the pre-period. Thirty-eight cancer drugs obtained some level of funding through the new process, but there was no significant difference in the distribution of decision types compared to the pre-reform period. Thematic analysis of patient and clinician input showed no clear relationship between issues raised and funding decision. Differences between SMC's and NICE's definitions of End-of-Life did not fully explain differences in funding decisions.
Conclusions: The Scottish Medicines Consortium's reforms have allowed funding of up to 38 cancer drugs that might previously have been rejected. However, the contribution of specific elements of the reforms to the final decision is unclear. The process could be improved by increased transparency in how the non-quantitative inputs influence decisions. Some disparities in funding decisions between England and Scotland are likely to remain despite recent process convergence.
Keywords: Access; Cancer; Cost-effectiveness; End of life; Funding; NICE; Orphan; Rare; Scottish medicines consortium.
Conflict of interest statement
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
LM, SW, HF and SR declare that they have no competing interests. RB is on the board of the biopharmaceutical company Celgene, and was Chair of the UK’s Precision Medicine Catapult.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
Similar articles
-
Access to orphan drugs in western Europe: can more systematic policymaking really help to avoid different decisions about the same drug?Expert Rev Pharmacoecon Outcomes Res. 2015;15(4):557-9. doi: 10.1586/14737167.2015.1045882. Epub 2015 May 14. Expert Rev Pharmacoecon Outcomes Res. 2015. PMID: 25973903
-
Clinical benefit, reimbursement outcomes, and prices of FDA-approved cancer drugs reviewed through Project Orbis in the USA, Canada, England, and Scotland: a retrospective, comparative analysis.Lancet Oncol. 2024 Aug;25(8):979-988. doi: 10.1016/S1470-2045(24)00286-9. Epub 2024 Jul 11. Lancet Oncol. 2024. PMID: 39004098
-
Economic Modeling Considerations for Rare Diseases.Value Health. 2018 May;21(5):515-524. doi: 10.1016/j.jval.2018.02.008. Epub 2018 Apr 17. Value Health. 2018. PMID: 29753347 Review.
-
Which factors enhance positive drug reimbursement recommendation in Scotland? A retrospective analysis 2006-2013.Value Health. 2015 Mar;18(2):284-91. doi: 10.1016/j.jval.2014.12.008. Epub 2015 Feb 4. Value Health. 2015. PMID: 25773564
-
Reimbursement of targeted cancer therapies within 3 different European health care systems.Clin Ther. 2015 Feb 1;37(2):474-80. doi: 10.1016/j.clinthera.2014.12.005. Epub 2015 Jan 28. Clin Ther. 2015. PMID: 25638534 Review.
Cited by
-
Cancer as the "perfect storm"? A qualitative study of public attitudes to health conditions.Health Sci Rep. 2017 Oct 27;1(1):e16. doi: 10.1002/hsr2.16. eCollection 2018 Jan. Health Sci Rep. 2017. PMID: 30024988 Free PMC article.
-
A Review of Rare Disease Policies and Orphan Drug Reimbursement Systems in 12 Eurasian Countries.Front Public Health. 2020 Jan 28;7:416. doi: 10.3389/fpubh.2019.00416. eCollection 2019. Front Public Health. 2020. PMID: 32117845 Free PMC article.
-
Systematic Literature Review of Access Pathways to Drugs for Patients with Rare Diseases.Appl Health Econ Health Policy. 2025 Mar;23(2):209-229. doi: 10.1007/s40258-024-00939-4. Epub 2024 Dec 28. Appl Health Econ Health Policy. 2025. PMID: 39731657
-
Lutetium oxodotreotide (177Lu-Dotatate) for the treatment of unresectable or metastatic progressive gastroenteropancreatic neuroendocrine tumors: a cost-effectiveness analysis for Scotland.BMC Cancer. 2021 Jan 5;21(1):10. doi: 10.1186/s12885-020-07710-7. BMC Cancer. 2021. PMID: 33402120 Free PMC article.
-
Appraisal of Novel Oncological Therapies by the Scottish Medicines Consortium and the National Institute for Health and Care Excellence: A Comparative Study of Six Years of Data.Cureus. 2023 Dec 15;15(12):e50560. doi: 10.7759/cureus.50560. eCollection 2023 Dec. Cureus. 2023. PMID: 38222244 Free PMC article.
References
-
- Chalkidou K. Evidence and values: paying for end-of-life drugs in the British NHS. Health Econ Policy Law. 2012;7:393–409. - PubMed
-
- NICE. Guide to the methods of technology appraisal. In. 2013. http://www.nice.org.uk/process/pmg9. - PubMed
-
- Scottish medicines consortium. SMC Modifiers used in Appraising New Medicines. In. 2012. http://www.scottishmedicines.org.uk/About_SMC/Policy_statements/SMC_Modi.... Accessed 1 Nov 2016.
-
- Scottish medicines consortium. A Guide to the Scottish medicines consortium. In. 2015. https://www.scottishmedicines.org.uk/About_SMC/What_we_do/SMC_Guide__web.... Accessed 7 Jul 2016.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

