Adipose stem cells from chronic pancreatitis patients improve mouse and human islet survival and function
- PMID: 28854965
- PMCID: PMC5577777
- DOI: 10.1186/s13287-017-0627-x
Adipose stem cells from chronic pancreatitis patients improve mouse and human islet survival and function
Abstract
Background: Chronic pancreatitis has surgical options including total pancreatectomy to control pain. To avoid surgical diabetes, the explanted pancreas can have islets harvested and transplanted. Immediately following total pancreatectomy with islet autotransplantation (TP-IAT), many islet cells die due to isolation and transplantation stresses. The percentage of patients remaining insulin free after TP-IAT is therefore low. We determined whether cotransplantation of adipose-derived mesenchymal stem cells (ASCs) from chronic pancreatitis patients (CP-ASCs) would protect islets after transplantation.
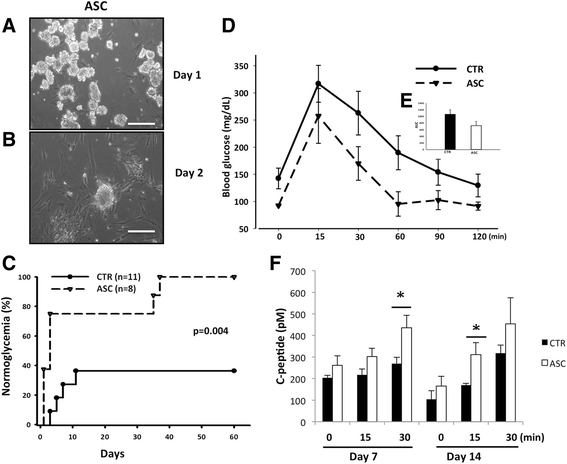
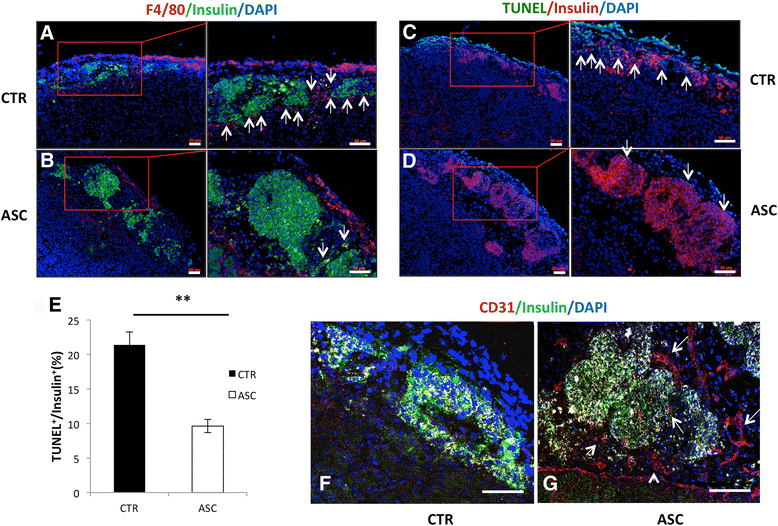
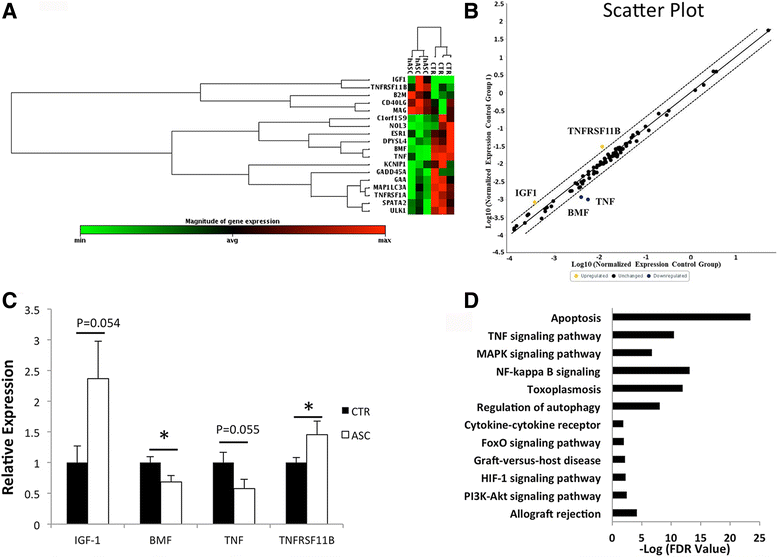
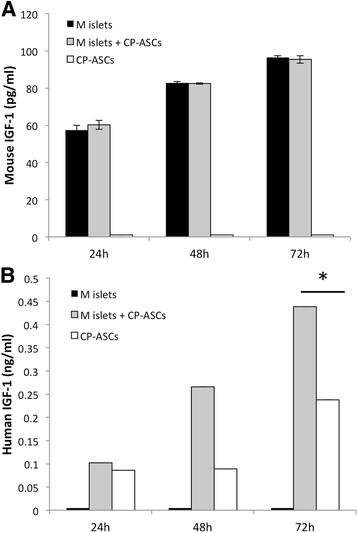
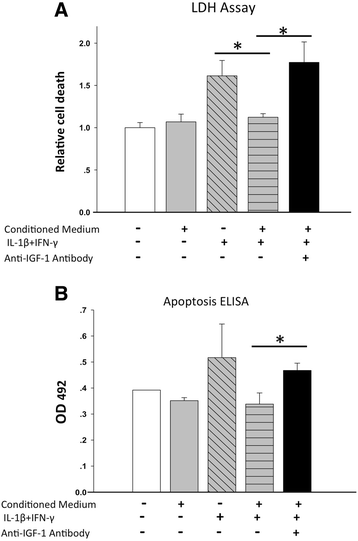
Methods: In a marginal mass islet transplantation model, islets from C57BL/6 mice were cotransplanted with CP-ASCs into syngeneic streptozotocin-treated diabetic mice. Treatment response was defined by the percentage of recipients reaching normoglycemia, and by the area under the curve for glucose and c-peptide in a glucose tolerance test. Macrophage infiltration, β-cell apoptosis, and islet graft vasculature were measured in transplanted islet grafts by immunohistochemistry. mRNA expression profiling of 84 apoptosis-related genes in islet grafts transplanted alone or with CP-ASCs was measured by the RT2 Profiler™ Apoptosis PCR Array. The impact of insulin-like growth factor-1 (IGF-1) on islet apoptosis was determined in islets stimulated with cytokines (IL-1β and IFN-γ) in the presence and absence of CP-ASC conditioned medium.
Results: CP-ASC-treated mice were more often normoglycemic compared to mice receiving islets alone. ASC cotransplantation reduced macrophage infiltration, β-cell death, suppressed expression of TNF-α and Bcl-2 modifying factor (BMF), and upregulated expressions of IGF-1 and TNF Receptor Superfamily Member 11b (TNFRSF11B) in islet grafts. Islets cultured in conditioned medium from CP-ASCs showed reduced cell death. This protective effect was diminished when IGF-1 was blocked in the conditioned medium by the anti-IGF-1 antibody.
Conclusion: Cotransplantation of islets with ASCs from the adipose of chronic pancreatitis patients improved islet survival and islet function after transplantation. The effects are in part mediated by paracrine secretion of IGF-1, suppression of inflammation, and promotion of angiogenesis. ASCs from chronic pancreatitis patients have the potential to be used as a synergistic therapy to enhance the efficacy of islet transplantation following pancreatectomy.
Keywords: Adipose stem cells; Chronic pancreatitis; Islet survival; Islet transplantation.
Conflict of interest statement
Ethics approval and consent to participate
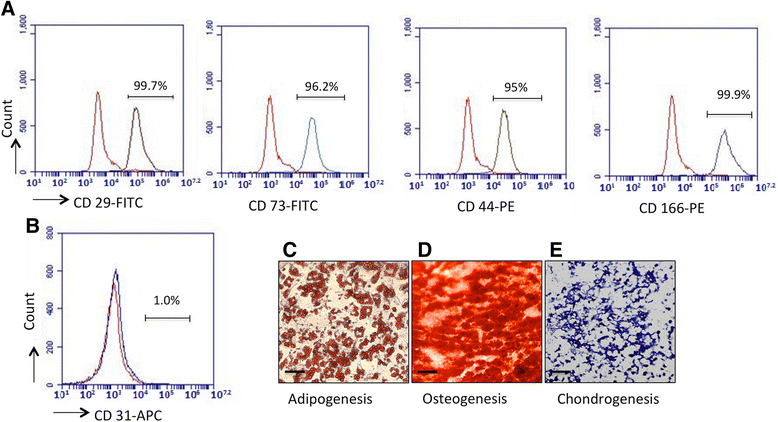
All mouse surgical procedures were approved by the Animal Care Committee at the Medical University of South Carolina (protocol #AR3055). ASCs were harvested from a small piece of fat tissue from chronic pancreatitis patients undergoing pancreatic surgery who gave informed consent for the study under protocols approved by the Medical University of South Carolina Internal Review Board (Pro00028011).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
-
- Dong H, Morgan K, Adams D, Wang H. Prevention of beta cell death in chronic pancreatitis. Adv Biosci Biotechnol. 2012;3(6A):782–7. doi: 10.4236/abb.2012.326098. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

